Below are the coefficients of association calculated by HealthCorrelator for Excel (HCE) for user John Doe. The coefficients of association are calculated as linear correlations in HCE (). The focus here is on the associations between fasting triglycerides and various other variables. Take a look at the coefficient of association at the top, with VLDL cholesterol, indicated with a red arrow. It is a very high 0.999.
Whoa! What is this – 0.999! Is John Doe a unique case? No, this strong association between fasting triglycerides and VLDL cholesterol is a very common pattern among HCE users. The reason is simple. VLDL cholesterol is not normally measured directly, but typically calculated based on fasting triglycerides, by dividing the fasting triglycerides measurement by 5. And there is an underlying reason for that - fasting triglycerides and VLDL cholesterol are actually very highly correlated, based on direct measurements of these two variables.
But if VLDL cholesterol is calculated based on fasting triglycerides (VLDL cholesterol = fasting triglycerides / 5), how come the correlation is 0.999, and not a perfect 1? The reason is the rounding error in the measurements. Whenever you see a correlation this high (i.e., 0.999), it is reasonable to suspect that the source is an underlying linear relationship disturbed by rounding error.
Fasting triglycerides are probably the most useful measures on standard lipid panels. For example, fasting triglycerides below 70 mg/dl suggest a pattern of LDL particles that is predominantly of large and buoyant particles. This pattern is associated with a low incidence of cardiovascular disease (). Also, chronically high fasting triglycerides are a well known marker of the metabolic syndrome, and a harbinger of type 2 diabetes.
Where do large and buoyant LDL particles come from? They frequently start as "big" (relatively speaking) blobs of fat, which are actually VLDL particles. The photo is from the excellent book by Elliott & Elliott (); it shows, on the same scale: (a) VLDL particles, (b) chylomicrons, (c) LDL particles, and (d) HDL particles. The dark bar at the bottom of each shot is 1000 A in length, or 100 nm (A = angstrom; nm = nanometer; 1 nm = 10 A).
If you consume an excessive amount of carbohydrates, my theory is that your liver will produce an abnormally large number of small VLDL particles (also shown on the photo above), a proportion of which will end up as small and dense LDL particles. The liver will do that relatively quickly, probably as a short-term compensatory mechanism to avoid glucose toxicity. It will essentially turn excess glucose, from excess carbohydrates, into fat. The VLDL particles carrying that fat in the form of triglycerides will be small because the liver will be in a hurry to clear the excess glucose in circulation, and will have no time to produce large particles, which take longer to produce individually.
This will end up leading to excess triglycerides hanging around in circulation, long after they should have been used as sources of energy. High fasting triglycerides will be a reflection of that. The graphs below, also generated by HCE for John Doe, show how fasting triglycerides and VLDL cholesterol vary in relation to refined carbohydrate consumption. Again, the graphs are not identical in shape because of rounding error; the shapes are almost identical.
Small and dense LDL particles, in the presence of other factors such as systemic inflammation, will contribute to the formation of atherosclerotic plaques. Again, the main source of these particles would be an excessive amount of carbohydrates. What is an excessive amount of carbohydrates? Generally speaking, it is an amount beyond your liver’s capacity to convert the resulting digestion byproducts, fructose and glucose, into liver glycogen. This may come from spaced consumption throughout the day, or acute consumption in an unnatural form (a can of regular coke), or both.
Liver glycogen is sugar stored in the liver. This is the main source of sugar for your brain. If your blood sugar levels become too low, your brain will get angry. Eventually it will go from angry to dead, and you will finally find out what awaits you in the afterlife.
Should you be a healthy athlete who severely depletes liver glycogen stores on a regular basis, you will probably have an above average liver glycogen storage and production capacity. That will be a result of long-term compensatory adaptation to glycogen depleting exercise (). As such, you may be able to consume large amounts of carbohydrates, and you will still not have high fasting triglycerides. You will not carry a lot of body fat either, because the carbohydrates will not be converted to fat and sent into circulation in VLDL particles. They will be used to make liver glycogen.
In fact, if you are a healthy athlete who severely depletes liver glycogen stores on a regular basis, excess calories will be just about the only thing that will contribute to body fat gain. Your threshold for “excess” carbohydrates will be so high that you will feel like the whole low carbohydrate community is not only misguided but also part of a conspiracy against people like you. If you are also an aggressive blog writer, you may feel compelled to tell the world something like this: “Here, I can eat 300 g of carbohydrates per day and maintain single-digit body fat levels! Take that you low carbohydrate idiots!”
Let us say you do not consume an excessive amount of carbohydrates; again, what is excessive or not varies, probably dramatically, from individual to individual. In this case your liver will produce a relatively small number of fat VLDL particles, which will end up as large and buoyant LDL particles. The fat in these large VLDL particles will likely not come primarily from conversion of glucose and/or fructose into fat (i.e., de novo lipogenesis), but from dietary sources of fat.
How do you avoid consuming excess carbohydrates? A good way of achieving that is to avoid man-made carbohydrate-rich foods. Another is adopting a low carbohydrate diet. Yet another is to become a healthy athlete who severely depletes liver glycogen stores on a regular basis; then you can eat a lot of bread, pasta, doughnuts and so on, and keep your fingers crossed for the future.
Either way, fasting triglycerides will be strongly correlated with VLDL cholesterol, because VLDL particles contain both triglycerides (“encapsulated” fat, not to be confused with “free” fatty acids) and cholesterol. If a large number of VLDL particles are produced by one’s liver, the person’s fasting triglycerides reading will be high. If a small number of VLDL particles are produced, even if they are fat particles, the fasting triglycerides reading will be relatively low. Neither VLDL cholesterol nor fasting triglycerides will be zero though.
Now, you may be wondering, how come a small number of fat VLDL particles will eventually lead to low fasting triglycerides? After all, they are fat particles, even though they occur in fewer numbers. My hypothesis is that having a large number of small-dense VLDL particles in circulation is an abnormal, unnatural state, and that our body is not well designed to deal with that state. Use of lipoprotein-bound fat as a source of energy in this state becomes somewhat less efficient, leading to high triglycerides in circulation; and also to hunger, as our mitochondria like fat.
This hypothesis, and the theory outlined above, fit well with the numbers I have been seeing for quite some time from HCE users. Note that it is a bit different from the more popular theory, particularly among low carbohydrate writers, that fat is force-stored in adipocytes (fat cells) by insulin and not released for use as energy, also leading to hunger. What I am saying here, which is compatible with this more popular theory, is that lipoproteins, like adipocytes, also end up holding more fat than they should if you consume excess carbohydrates, and for longer.
Want to improve your health? Consider replacing things like bread and cereal with butter and eggs in your diet (). And also go see you doctor (); if he disagrees with this recommendation, ask him to read this post and explain why he disagrees.
Healthy living soul there is a strong, here are a few lots of information about health. Some information on body care, skin care, eye health, and others.
Showing posts with label LDL. Show all posts
Showing posts with label LDL. Show all posts
Monday, November 28, 2011
Monday, June 13, 2011
Alcohol intake increases LDL cholesterol, in some people
Occasionally I get emails from people experiencing odd fluctuations in health markers, and trying to figure out what is causing those fluctuations. Spikes in LDL cholesterol without any change in diet seem to be a common occurrence, especially in men.
LDL cholesterol is a reflection of many things. It is one of the least useful measures in standard lipid profiles, as a predictor of future health problems. Nevertheless, if one’s diet is not changing, whether it is high or low in fat, significant fluctuations in LDL cholesterol may signal a change in inflammatory status. Generally speaking, the more systemic inflammation, the higher is the measured LDL cholesterol.
Corella and colleagues (2001) looked into alcohol consumption and its effect on LDL cholesterol, as part of the Framingham Offspring Study. They split the data into three genotypes, which are allele combinations. Alleles are genes variations; that is, they are variations in the sections of DNA that have been identified as coding for observable traits. The table below summarizes what they have found. Take a look at the last two columns on the right.
As you can see, for men with the E2 genotype, alcohol consumption significantly decreases LDL cholesterol. For men with the E4 genotype, alcohol consumption significantly increases LDL cholesterol. No significant effects were observed in women. The figure below illustrates the magnitude of the effects observed in men.
On average, alcohol consumption was moderate, around 15 g per day, and did not vary significantly based on genotype. This is important. Otherwise one could argue that a particular genotype predisposed individuals to drink more, which would be a major confounder in this study. Other confounders were also ruled out through multivariate controls - e.g., fat and calorie intake, and smoking.
Alcohol consumption in moderation seems, on average, to be beneficial. But for some individuals, particularly men with a certain genotype, it may be advisable to completely abstain from alcohol consumption. Who are those folks? They are the ones for whom LDL cholesterol goes up significantly following moderate alcohol consumption.
LDL cholesterol is a reflection of many things. It is one of the least useful measures in standard lipid profiles, as a predictor of future health problems. Nevertheless, if one’s diet is not changing, whether it is high or low in fat, significant fluctuations in LDL cholesterol may signal a change in inflammatory status. Generally speaking, the more systemic inflammation, the higher is the measured LDL cholesterol.
Corella and colleagues (2001) looked into alcohol consumption and its effect on LDL cholesterol, as part of the Framingham Offspring Study. They split the data into three genotypes, which are allele combinations. Alleles are genes variations; that is, they are variations in the sections of DNA that have been identified as coding for observable traits. The table below summarizes what they have found. Take a look at the last two columns on the right.
As you can see, for men with the E2 genotype, alcohol consumption significantly decreases LDL cholesterol. For men with the E4 genotype, alcohol consumption significantly increases LDL cholesterol. No significant effects were observed in women. The figure below illustrates the magnitude of the effects observed in men.
On average, alcohol consumption was moderate, around 15 g per day, and did not vary significantly based on genotype. This is important. Otherwise one could argue that a particular genotype predisposed individuals to drink more, which would be a major confounder in this study. Other confounders were also ruled out through multivariate controls - e.g., fat and calorie intake, and smoking.
Alcohol consumption in moderation seems, on average, to be beneficial. But for some individuals, particularly men with a certain genotype, it may be advisable to completely abstain from alcohol consumption. Who are those folks? They are the ones for whom LDL cholesterol goes up significantly following moderate alcohol consumption.
Monday, May 30, 2011
Interview with Jimmy Moore, and basics of intima-media thickness and plaque tests
Let me start this post by telling you that my interview with Jimmy Moore is coming up in about a week. Jimmy and I talk about evolution, statistics, and health – the main themes of this blog. We talk also about other things, and probably do not agree on everything. The interview was actually done a while ago, so I don’t remember exactly what we discussed.
From what I remember from mine and other interviews (I listen to Jimmy's podcasts regularly), I think I am the guest who has mentioned the most people during an interview – Gary Taubes, Chris Masterjohn, Carbsane, Petro (a.k.a., Peter “the Hyperlipid”), T. Colin Campbell, Denise Minger, Kurt Harris, Stephan Guyenet, Art De Vany, and a few others. What was I thinking?
In case you listen and wonder, my accent is a mix of Brazilian Portuguese, New Zealand English (where I am called “Need”), American English, and the dialect spoken in the “country” of Texas. The strongest influences are probably American English and Brazilian Portuguese.
Anyway, when medical doctors (MDs) look at someone’s lipid panel, one single number tends to draw their attention: the LDL cholesterol. That is essentially the amount of cholesterol in LDL particles.
One’s LDL cholesterol is a reflection of many factors, including: diet, amount of cholesterol produced by the liver, amount of cholesterol actually used by your body, amount of cholesterol recycled by the liver, and level of systemic inflammation. This number is usually calculated, and often very different from the number you get through a VAP test.
It is not uncommon for a high saturated fat diet to lead to a benign increase in LDL cholesterol. In this case the LDL particles will be large, which will also be reflected in a low “fasting triglycerides number” (lower than 70 mg/dl). While I say "benign" here, which implies a neutral effect on health, an increase in LDL cholesterol in this context may actually be health promoting.
Large LDL particles are less likely to cross the gaps in the endothelium, the thin layer of cells that lines the interior surface of blood vessels, and form atheromatous plaques.
Still, when an MD sees an LDL cholesterol higher than 100 mg/dl, more often than not he or she will tell you that it is bad news. Whether that is bad news or not is really speculation, even for high LDL numbers. A more reliable approach is to check one’s arteries directly. Interestingly, atheromatous plaques only form in arteries, not in veins.
The figure below (from: Novogen.com) shows a photomicrograph of carotid arteries from rabbits, which are very similar, qualitatively speaking, to those of humans. The meanings of the letters are: L = lumen; I = intima; M = media; and A = adventitia. The one on the right has significantly lower intima-media (I-M) thickness than the one on the left.
Atherosclerosis in humans tends to lead to an increase in I-M thickness; the I-M area being normally where atheromatous plaques grow. Aging also leads to an increase in I-M thickness. Typically one’s risk of premature death from cardiovascular complications correlates with one’s I-M thickness’ “distance” from that of low-risk individuals in the same sex and age group.
This notion has led to the coining of the term “vascular age”. For example, someone may be 30 years old, but have a vascular age of 80, meaning that his or her I-M thickness is that of an average 80-year-old. Conversely, someone may be 80 and have a vascular age of 30.
Nearly everybody’s I-M thickness goes up with age, even people who live to be 100 or more. Incidentally, this is true for average blood glucose levels as well. In long-living people they both go up slowly.
I-M thickness tests are noninvasive, based on external ultrasound, and often covered by health insurance. They take only a few minutes to conduct. Their reports provide information about one’s I-M thickness and its relative position in the same sex and age group, as well as the amount of deposited plaque. The latter is frequently provided as a bonus, since it can also be inferred with reasonable precision from the computer images generated via ultrasound.
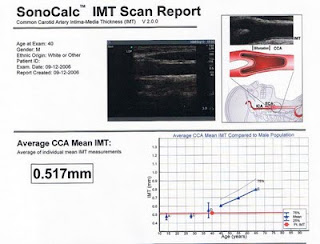
Below is the top part of a typical I-M thickness test report (from: Sonosite.com). It shows a person’s average (or mean) I-M thickness; the red dot on the graph. The letter notations (A … E) are for reference groups. For the majority of the folks doing this test, the most important on this report are the thick and thin lines indicated as E, which are based on Aminbakhsh and Mancini’s (1999) study.
The reason why the thick and thin lines indicated as E are the most important for the majority of folks taking this test is that they are based on a study that provides one of the best reference ranges for people who are 45 and older, who are usually the ones getting their I-M thickness tested. Roughly speaking, if your red dot is above the thin line, you are at increased risk of cardiovascular disease.
Most people will fall in between the thick and thin lines. Those below the thick line (with the little blue triangles) are at very low risk, especially if they have little to no plaque. The person for whom this test was made is at very low risk. His red dot is below the thick line, when that line is extended to the little triangle indicated as D.
Below is the bottom part of the I-M thickness test report. The max I-M thickness score shown here tends to add little in terms of diagnosis to the mean score shown earlier. Here the most important part is the summary, under “Comments”. It says that the person has no plaque, and is at a lower risk of heart attack. If you do an I-M thickness test, your doctor will probably be able to tell you more about these results.
I like numbers, so I had an I-M thickness test done recently on me. When the doctor saw the results, which we discussed, he told me that he could guarantee two things: (1) I would die; and (2) but not of heart disease. MDs have an interesting sense of humor; just hang out with a group of them during a “happy hour” and you’ll see.
My red dot was below the thick line, and I had a plaque measurement of zero. I am 47 years old, eat about 1 lb of meat per day, and around 20 eggs per week - with the yolk. About half of the meat I eat comes from animal organs (mostly liver) and seafood. I eat organ meats about once a week, and seafood three times a week. This is an enormous amount of dietary cholesterol, by American diet standards. My saturated fat intake is also high by the same standards.
You can check the post on my transformation to see what I have been doing for years now, and some of the results in terms of levels of energy, disease, and body fat levels. Keep in mind that mine are essentially the results of a single-individual experiment; results that clearly contradict the lipid hypothesis. Still, they are also consistent with a lot of fairly reliable empirical research.
From what I remember from mine and other interviews (I listen to Jimmy's podcasts regularly), I think I am the guest who has mentioned the most people during an interview – Gary Taubes, Chris Masterjohn, Carbsane, Petro (a.k.a., Peter “the Hyperlipid”), T. Colin Campbell, Denise Minger, Kurt Harris, Stephan Guyenet, Art De Vany, and a few others. What was I thinking?
In case you listen and wonder, my accent is a mix of Brazilian Portuguese, New Zealand English (where I am called “Need”), American English, and the dialect spoken in the “country” of Texas. The strongest influences are probably American English and Brazilian Portuguese.
Anyway, when medical doctors (MDs) look at someone’s lipid panel, one single number tends to draw their attention: the LDL cholesterol. That is essentially the amount of cholesterol in LDL particles.
One’s LDL cholesterol is a reflection of many factors, including: diet, amount of cholesterol produced by the liver, amount of cholesterol actually used by your body, amount of cholesterol recycled by the liver, and level of systemic inflammation. This number is usually calculated, and often very different from the number you get through a VAP test.
It is not uncommon for a high saturated fat diet to lead to a benign increase in LDL cholesterol. In this case the LDL particles will be large, which will also be reflected in a low “fasting triglycerides number” (lower than 70 mg/dl). While I say "benign" here, which implies a neutral effect on health, an increase in LDL cholesterol in this context may actually be health promoting.
Large LDL particles are less likely to cross the gaps in the endothelium, the thin layer of cells that lines the interior surface of blood vessels, and form atheromatous plaques.
Still, when an MD sees an LDL cholesterol higher than 100 mg/dl, more often than not he or she will tell you that it is bad news. Whether that is bad news or not is really speculation, even for high LDL numbers. A more reliable approach is to check one’s arteries directly. Interestingly, atheromatous plaques only form in arteries, not in veins.
The figure below (from: Novogen.com) shows a photomicrograph of carotid arteries from rabbits, which are very similar, qualitatively speaking, to those of humans. The meanings of the letters are: L = lumen; I = intima; M = media; and A = adventitia. The one on the right has significantly lower intima-media (I-M) thickness than the one on the left.
Atherosclerosis in humans tends to lead to an increase in I-M thickness; the I-M area being normally where atheromatous plaques grow. Aging also leads to an increase in I-M thickness. Typically one’s risk of premature death from cardiovascular complications correlates with one’s I-M thickness’ “distance” from that of low-risk individuals in the same sex and age group.
This notion has led to the coining of the term “vascular age”. For example, someone may be 30 years old, but have a vascular age of 80, meaning that his or her I-M thickness is that of an average 80-year-old. Conversely, someone may be 80 and have a vascular age of 30.
Nearly everybody’s I-M thickness goes up with age, even people who live to be 100 or more. Incidentally, this is true for average blood glucose levels as well. In long-living people they both go up slowly.
I-M thickness tests are noninvasive, based on external ultrasound, and often covered by health insurance. They take only a few minutes to conduct. Their reports provide information about one’s I-M thickness and its relative position in the same sex and age group, as well as the amount of deposited plaque. The latter is frequently provided as a bonus, since it can also be inferred with reasonable precision from the computer images generated via ultrasound.
Below is the top part of a typical I-M thickness test report (from: Sonosite.com). It shows a person’s average (or mean) I-M thickness; the red dot on the graph. The letter notations (A … E) are for reference groups. For the majority of the folks doing this test, the most important on this report are the thick and thin lines indicated as E, which are based on Aminbakhsh and Mancini’s (1999) study.
The reason why the thick and thin lines indicated as E are the most important for the majority of folks taking this test is that they are based on a study that provides one of the best reference ranges for people who are 45 and older, who are usually the ones getting their I-M thickness tested. Roughly speaking, if your red dot is above the thin line, you are at increased risk of cardiovascular disease.
Most people will fall in between the thick and thin lines. Those below the thick line (with the little blue triangles) are at very low risk, especially if they have little to no plaque. The person for whom this test was made is at very low risk. His red dot is below the thick line, when that line is extended to the little triangle indicated as D.
Below is the bottom part of the I-M thickness test report. The max I-M thickness score shown here tends to add little in terms of diagnosis to the mean score shown earlier. Here the most important part is the summary, under “Comments”. It says that the person has no plaque, and is at a lower risk of heart attack. If you do an I-M thickness test, your doctor will probably be able to tell you more about these results.
I like numbers, so I had an I-M thickness test done recently on me. When the doctor saw the results, which we discussed, he told me that he could guarantee two things: (1) I would die; and (2) but not of heart disease. MDs have an interesting sense of humor; just hang out with a group of them during a “happy hour” and you’ll see.
My red dot was below the thick line, and I had a plaque measurement of zero. I am 47 years old, eat about 1 lb of meat per day, and around 20 eggs per week - with the yolk. About half of the meat I eat comes from animal organs (mostly liver) and seafood. I eat organ meats about once a week, and seafood three times a week. This is an enormous amount of dietary cholesterol, by American diet standards. My saturated fat intake is also high by the same standards.
You can check the post on my transformation to see what I have been doing for years now, and some of the results in terms of levels of energy, disease, and body fat levels. Keep in mind that mine are essentially the results of a single-individual experiment; results that clearly contradict the lipid hypothesis. Still, they are also consistent with a lot of fairly reliable empirical research.
Wednesday, September 8, 2010
The China Study II: Cholesterol seems to protect against cardiovascular disease
First of all, many thanks are due to Dr. Campbell and his collaborators for collecting and compiling the data used in this analysis. This data is from this site, created by those researchers to disseminate the data from a study often referred to as the “China Study II”. It has already been analyzed by other bloggers. Notable analyses have been conducted by Ricardo at Canibais e Reis, Stan at Heretic, and Denise at Raw Food SOS.
The analyses in this post differ from those other analyses in various aspects. One of them is that data for males and females were used separately for each county, instead of the totals per county. Only two data points per county were used (for males and females). This increased the sample size of the dataset without artificially reducing variance (for more details, see “Notes” at the end of the post), which is desirable since the dataset is relatively small. This also allowed for the test of commonsense assumptions (e.g., the protective effects of being female), which is always a good idea in a complex analysis because violation of commonsense assumption may suggest data collection or analysis error. On the other hand, it required the inclusion of a sex variable as a control variable in the analysis, which is no big deal.
The analysis was conducted using WarpPLS. Below is the model with the main results of the analysis. (Click on it to enlarge. Use the "CRTL" and "+" keys to zoom in, and CRTL" and "-" to zoom out.) The arrows explore associations between variables, which are shown within ovals. The meaning of each variable is the following: SexM1F2 = sex, with 1 assigned to males and 2 to females; HDLCHOL = HDL cholesterol; TOTCHOL = total cholesterol; MSCHIST = mortality from schistosomiasis infection; and MVASC = mortality from all cardiovascular diseases.
The variables to the left of MVASC are the main predictors of interest in the model – HDLCHOL and TOTCHOL. The ones to the right are control variables – SexM1F2 and MSCHIST. The path coefficients (indicated as beta coefficients) reflect the strength of the relationships. A negative beta means that the relationship is negative; i.e., an increase in a variable is associated with a decrease in the variable that it points to. The P values indicate the statistical significance of the relationship; a P lower than 0.05 generally means a significant relationship (95 percent or higher likelihood that the relationship is “real”).
In summary, this is what the model above is telling us:
- As HDL cholesterol increases, total cholesterol increases significantly (beta=0.48; P<0.01). This is to be expected, as HDL is a main component of total cholesterol, together with VLDL and LDL cholesterol.
- As total cholesterol increases, mortality from all cardiovascular diseases decreases significantly (beta=-0.25; P<0.01). This is to be expected if we assume that total cholesterol is in part an intervening variable between HDL cholesterol and mortality from all cardiovascular diseases. This assumption can be tested through a separate model (more below). Also, there is more to this story, as noted below.
- The effect of HDL cholesterol on mortality from all cardiovascular diseases is insignificant when we control for the effect of total cholesterol (beta=-0.08; P=0.26). This suggests that HDL’s protective role is subsumed by the variable total cholesterol, and also that it is possible that there is something else associated with total cholesterol that makes it protective. Otherwise the effect of total cholesterol might have been insignificant, and the effect of HDL cholesterol significant (the reverse of what we see here).
- Being female is significantly associated with a reduction in mortality from all cardiovascular diseases (beta=-0.16; P=0.01). This is to be expected. In other words, men are women with a few design flaws. (This situation reverses itself a bit after menopause.)
- Mortality from schistosomiasis infection is significantly and inversely associated with mortality from all cardiovascular diseases (beta=-0.28; P<0.01). This is probably due to those dying from schistosomiasis infection not being entered in the dataset as dying from cardiovascular diseases, and vice-versa.
Two other main components of total cholesterol, in addition to HDL cholesterol, are VLDL and LDL cholesterol. These are carried in particles, known as lipoproteins. VLDL cholesterol is usually represented as a fraction of triglycerides in cholesterol equations (e.g., the Friedewald and Iranian equations). It usually correlates inversely with HDL; that is, as HDL cholesterol increases, usually VLDL cholesterol decreases. Given this and the associations discussed above, it seems that LDL cholesterol is a good candidate for the possible “something else associated with total cholesterol that makes it protective”. But waidaminet! Is it possible that the demon particle, the LDL, serves any purpose other than giving us heart attacks?
The graph below shows the shape of the association between total cholesterol (TOTCHOL) and mortality from all cardiovascular diseases (MVASC). The values are provided in standardized format; e.g., 0 is the average, 1 is one standard deviation above the mean, and so on. The curve is the best-fitting S curve obtained by the software (an S curve is a slightly more complex curve than a U curve).
The graph below shows some of the data in unstandardized format, and organized differently. The data is grouped here in ranges of total cholesterol, which are shown on the horizontal axis. The lowest and highest ranges in the dataset are shown, to highlight the magnitude of the apparently protective effect. Here the two variables used to calculate mortality from all cardiovascular diseases (MVASC; see “Notes” at the end of this post) were added. Clearly the lowest mortality from all cardiovascular diseases is in the highest total cholesterol range, 172.5 to 180; and the highest mortality in the lowest total cholesterol range, 120 to 127.5. The difference is quite large; the mortality in the lowest range is approximately 3.3 times higher than in the highest.
The shape of the S-curve graph above suggests that there are other variables that are confounding the results a bit. Mortality from all cardiovascular diseases does seem to generally go down with increases in total cholesterol, but the smooth inflection point at the middle of the S-curve graph suggests a more complex variation pattern that may be influenced by other variables (e.g., smoking, dietary patterns, or even schistosomiasis infection; see “Notes” at the end of this post).
As mentioned before, total cholesterol is strongly influenced by HDL cholesterol, so below is the model with only HDL cholesterol (HDLCHOL) pointing at mortality from all cardiovascular diseases (MVASC), and the control variable sex (SexM1F2).
The graph above confirms the assumption that HDL’s protective role is subsumed by the variable total cholesterol. When the variable total cholesterol is removed from the model, as it was done above, the protective effect of HDL cholesterol becomes significant (beta=-0.27; P<0.01). The control variable sex (SexM1F2) was retained even in this targeted HDL effect model because of the expected confounding effect of sex; females generally tend to have higher HDL cholesterol and less cardiovascular disease than males.
Below, in the “Notes” section (after the “Reference”) are several notes, some of which are quite technical. Providing them separately hopefully has made the discussion above a bit easier to follow. The notes also point at some limitations of the analysis. This data needs to be analyzed from different angles, using multiple models, so that firmer conclusions can be reached. Still, the overall picture that seems to be emerging is at odds with previous beliefs based on the same dataset.
What could be increasing the apparently protective HDL and total cholesterol in this dataset? High consumption of animal foods, particularly foods rich in saturated fat and cholesterol, are strong candidates. Low consumption of vegetable oils rich in linoleic acid, and of foods rich in refined carbohydrates, are also good candidates. Maybe it is a combination of these.
We need more analyses!
Reference:
Kock, N. (2010). WarpPLS 1.0 User Manual. Laredo, Texas: ScriptWarp Systems.
Notes:
- The path coefficients (indicated as beta coefficients) reflect the strength of the relationships; they are a bit like standard univariate (or Pearson) correlation coefficients, except that they take into consideration multivariate relationships (they control for competing effects on each variable).
- The R-squared values reflect the percentage of explained variance for certain variables; the higher they are, the better the model fit with the data. In complex and multi-factorial phenomena such as health-related phenomena, many would consider an R-squared of 0.20 as acceptable. Still, such an R-squared would mean that 80 percent of the variance for a particularly variable is unexplained by the data.
- The P values have been calculated using a nonparametric technique, a form of resampling called jackknifing, which does not require the assumption that the data is normally distributed to be met. This and other related techniques also tend to yield more reliable results for small samples, and samples with outliers (as long as the outliers are “good” data, and are not the result of measurement error).
- Colinearity is an important consideration in models that analyze the effect of multiple predictors on one single variable. This is particularly true for multiple regression models, where there is a temptation of adding many predictors to the model to see which ones come out as the “winners”. This often backfires, as colinearity can severely distort the results. Some multiple regression techniques, such as automated stepwise regression with backward elimination, are particularly vulnerable to this problem. Colinearity is not the same as correlation, and thus is defined and measured differently. Two predictor variables may be significantly correlated and still have low colinearity. A reasonably reliable measure of colinearity is the variance inflation factor. Colinearity was tested in this model, and was found to be low.
- An effort was made here to avoid multiple data points per county (even though this was available for some variables), because this could artificially reduce the variance for each variable, and potentially bias the results. The reason for this is that multiple answers from a single county would normally be somewhat correlated; a higher degree of intra-county correlation than inter-county correlation. The resulting bias would be difficult to control for, via one or more control variables. With only two data points per county, one for males and the other for females, one can control for intra-country correlation by adding a “dummy” sex variable to the analysis, as a control variable. This was done here.
- Mortality from schistosomiasis infection (MSCHIST) is a variable that tends to affect the results in a way that makes it more difficult to make sense of them. Generally this is true for any infectious diseases that significantly affect a population under study. The problem with infection is that people with otherwise good health or habits may get the infection, and people with bad health and habits may not. Since cholesterol is used by the human body to fight disease, it may go up, giving the impression that it is going up for some other reason. Perhaps instead of controlling for its effect, as done here, it would have been better to remove from the analysis those counties with deaths from schistosomiasis infection. (See also this post, and this one.)
- Different parts of the data were collected at different times. It seems that the mortality data is for the period 1986-88, and the rest of the data is for 1989. This may have biased the results somewhat, even though the time lag is not that long, especially if there were changes in certain health trends from one period to the other. For example, major migrations from one county to another could have significantly affected the results.
- The following measures were used, from this online dataset like the other measures. P002 HDLCHOL, for HDLCHOL; P001 TOTCHOL, for TOTCHOL; and M021 SCHISTOc, for MSCHIST.
- SexM1F2 is a “dummy” variable that was coded with 1 assigned to males and 2 to females. As such, it essentially measures the “degree of femaleness” of the respondents. Being female is generally protective against cardiovascular disease, a situation that reverts itself a bit after menopause.
- MVASC is a composite measure of the two following variables, provided as component measures of mortality from all cardiovascular diseases: M058 ALLVASCb (ages 0-34), and M059 ALLVASCc (ages 35-69). A couple of obvious problems: (a) they does not include data on people older than 69; and (b) they seem to capture a lot of diseases, including some that do not seem like typical cardiovascular diseases. A factor analysis was conducted, and the loadings and cross-loadings suggested good validity. Composite reliability was also good. So essentially MVASC is measured here as a “latent variable” with two “indicators”. Why do this? The reason is that it reduces the biasing effects of incomplete data and measurement error (e.g., exclusion of folks older than 69). By the way, there is always some measurement error in any dataset.
- This note is related to measurement error in connection with the indicators for MVASC. There is something odd about the variables M058 ALLVASCb (ages 0-34), and M059 ALLVASCc (ages 35-69). According to the dataset, mortality from cardiovascular diseases for ages 0-34 is typically higher than for 35-69, for many counties. Given the good validity and reliability for MVASC as a latent variable, it is possible that the values for these two indicator variables were simply swapped by mistake.
The analyses in this post differ from those other analyses in various aspects. One of them is that data for males and females were used separately for each county, instead of the totals per county. Only two data points per county were used (for males and females). This increased the sample size of the dataset without artificially reducing variance (for more details, see “Notes” at the end of the post), which is desirable since the dataset is relatively small. This also allowed for the test of commonsense assumptions (e.g., the protective effects of being female), which is always a good idea in a complex analysis because violation of commonsense assumption may suggest data collection or analysis error. On the other hand, it required the inclusion of a sex variable as a control variable in the analysis, which is no big deal.
The analysis was conducted using WarpPLS. Below is the model with the main results of the analysis. (Click on it to enlarge. Use the "CRTL" and "+" keys to zoom in, and CRTL" and "-" to zoom out.) The arrows explore associations between variables, which are shown within ovals. The meaning of each variable is the following: SexM1F2 = sex, with 1 assigned to males and 2 to females; HDLCHOL = HDL cholesterol; TOTCHOL = total cholesterol; MSCHIST = mortality from schistosomiasis infection; and MVASC = mortality from all cardiovascular diseases.
The variables to the left of MVASC are the main predictors of interest in the model – HDLCHOL and TOTCHOL. The ones to the right are control variables – SexM1F2 and MSCHIST. The path coefficients (indicated as beta coefficients) reflect the strength of the relationships. A negative beta means that the relationship is negative; i.e., an increase in a variable is associated with a decrease in the variable that it points to. The P values indicate the statistical significance of the relationship; a P lower than 0.05 generally means a significant relationship (95 percent or higher likelihood that the relationship is “real”).
In summary, this is what the model above is telling us:
- As HDL cholesterol increases, total cholesterol increases significantly (beta=0.48; P<0.01). This is to be expected, as HDL is a main component of total cholesterol, together with VLDL and LDL cholesterol.
- As total cholesterol increases, mortality from all cardiovascular diseases decreases significantly (beta=-0.25; P<0.01). This is to be expected if we assume that total cholesterol is in part an intervening variable between HDL cholesterol and mortality from all cardiovascular diseases. This assumption can be tested through a separate model (more below). Also, there is more to this story, as noted below.
- The effect of HDL cholesterol on mortality from all cardiovascular diseases is insignificant when we control for the effect of total cholesterol (beta=-0.08; P=0.26). This suggests that HDL’s protective role is subsumed by the variable total cholesterol, and also that it is possible that there is something else associated with total cholesterol that makes it protective. Otherwise the effect of total cholesterol might have been insignificant, and the effect of HDL cholesterol significant (the reverse of what we see here).
- Being female is significantly associated with a reduction in mortality from all cardiovascular diseases (beta=-0.16; P=0.01). This is to be expected. In other words, men are women with a few design flaws. (This situation reverses itself a bit after menopause.)
- Mortality from schistosomiasis infection is significantly and inversely associated with mortality from all cardiovascular diseases (beta=-0.28; P<0.01). This is probably due to those dying from schistosomiasis infection not being entered in the dataset as dying from cardiovascular diseases, and vice-versa.
Two other main components of total cholesterol, in addition to HDL cholesterol, are VLDL and LDL cholesterol. These are carried in particles, known as lipoproteins. VLDL cholesterol is usually represented as a fraction of triglycerides in cholesterol equations (e.g., the Friedewald and Iranian equations). It usually correlates inversely with HDL; that is, as HDL cholesterol increases, usually VLDL cholesterol decreases. Given this and the associations discussed above, it seems that LDL cholesterol is a good candidate for the possible “something else associated with total cholesterol that makes it protective”. But waidaminet! Is it possible that the demon particle, the LDL, serves any purpose other than giving us heart attacks?
The graph below shows the shape of the association between total cholesterol (TOTCHOL) and mortality from all cardiovascular diseases (MVASC). The values are provided in standardized format; e.g., 0 is the average, 1 is one standard deviation above the mean, and so on. The curve is the best-fitting S curve obtained by the software (an S curve is a slightly more complex curve than a U curve).
The graph below shows some of the data in unstandardized format, and organized differently. The data is grouped here in ranges of total cholesterol, which are shown on the horizontal axis. The lowest and highest ranges in the dataset are shown, to highlight the magnitude of the apparently protective effect. Here the two variables used to calculate mortality from all cardiovascular diseases (MVASC; see “Notes” at the end of this post) were added. Clearly the lowest mortality from all cardiovascular diseases is in the highest total cholesterol range, 172.5 to 180; and the highest mortality in the lowest total cholesterol range, 120 to 127.5. The difference is quite large; the mortality in the lowest range is approximately 3.3 times higher than in the highest.
The shape of the S-curve graph above suggests that there are other variables that are confounding the results a bit. Mortality from all cardiovascular diseases does seem to generally go down with increases in total cholesterol, but the smooth inflection point at the middle of the S-curve graph suggests a more complex variation pattern that may be influenced by other variables (e.g., smoking, dietary patterns, or even schistosomiasis infection; see “Notes” at the end of this post).
As mentioned before, total cholesterol is strongly influenced by HDL cholesterol, so below is the model with only HDL cholesterol (HDLCHOL) pointing at mortality from all cardiovascular diseases (MVASC), and the control variable sex (SexM1F2).
The graph above confirms the assumption that HDL’s protective role is subsumed by the variable total cholesterol. When the variable total cholesterol is removed from the model, as it was done above, the protective effect of HDL cholesterol becomes significant (beta=-0.27; P<0.01). The control variable sex (SexM1F2) was retained even in this targeted HDL effect model because of the expected confounding effect of sex; females generally tend to have higher HDL cholesterol and less cardiovascular disease than males.
Below, in the “Notes” section (after the “Reference”) are several notes, some of which are quite technical. Providing them separately hopefully has made the discussion above a bit easier to follow. The notes also point at some limitations of the analysis. This data needs to be analyzed from different angles, using multiple models, so that firmer conclusions can be reached. Still, the overall picture that seems to be emerging is at odds with previous beliefs based on the same dataset.
What could be increasing the apparently protective HDL and total cholesterol in this dataset? High consumption of animal foods, particularly foods rich in saturated fat and cholesterol, are strong candidates. Low consumption of vegetable oils rich in linoleic acid, and of foods rich in refined carbohydrates, are also good candidates. Maybe it is a combination of these.
We need more analyses!
Reference:
Kock, N. (2010). WarpPLS 1.0 User Manual. Laredo, Texas: ScriptWarp Systems.
Notes:
- The path coefficients (indicated as beta coefficients) reflect the strength of the relationships; they are a bit like standard univariate (or Pearson) correlation coefficients, except that they take into consideration multivariate relationships (they control for competing effects on each variable).
- The R-squared values reflect the percentage of explained variance for certain variables; the higher they are, the better the model fit with the data. In complex and multi-factorial phenomena such as health-related phenomena, many would consider an R-squared of 0.20 as acceptable. Still, such an R-squared would mean that 80 percent of the variance for a particularly variable is unexplained by the data.
- The P values have been calculated using a nonparametric technique, a form of resampling called jackknifing, which does not require the assumption that the data is normally distributed to be met. This and other related techniques also tend to yield more reliable results for small samples, and samples with outliers (as long as the outliers are “good” data, and are not the result of measurement error).
- Colinearity is an important consideration in models that analyze the effect of multiple predictors on one single variable. This is particularly true for multiple regression models, where there is a temptation of adding many predictors to the model to see which ones come out as the “winners”. This often backfires, as colinearity can severely distort the results. Some multiple regression techniques, such as automated stepwise regression with backward elimination, are particularly vulnerable to this problem. Colinearity is not the same as correlation, and thus is defined and measured differently. Two predictor variables may be significantly correlated and still have low colinearity. A reasonably reliable measure of colinearity is the variance inflation factor. Colinearity was tested in this model, and was found to be low.
- An effort was made here to avoid multiple data points per county (even though this was available for some variables), because this could artificially reduce the variance for each variable, and potentially bias the results. The reason for this is that multiple answers from a single county would normally be somewhat correlated; a higher degree of intra-county correlation than inter-county correlation. The resulting bias would be difficult to control for, via one or more control variables. With only two data points per county, one for males and the other for females, one can control for intra-country correlation by adding a “dummy” sex variable to the analysis, as a control variable. This was done here.
- Mortality from schistosomiasis infection (MSCHIST) is a variable that tends to affect the results in a way that makes it more difficult to make sense of them. Generally this is true for any infectious diseases that significantly affect a population under study. The problem with infection is that people with otherwise good health or habits may get the infection, and people with bad health and habits may not. Since cholesterol is used by the human body to fight disease, it may go up, giving the impression that it is going up for some other reason. Perhaps instead of controlling for its effect, as done here, it would have been better to remove from the analysis those counties with deaths from schistosomiasis infection. (See also this post, and this one.)
- Different parts of the data were collected at different times. It seems that the mortality data is for the period 1986-88, and the rest of the data is for 1989. This may have biased the results somewhat, even though the time lag is not that long, especially if there were changes in certain health trends from one period to the other. For example, major migrations from one county to another could have significantly affected the results.
- The following measures were used, from this online dataset like the other measures. P002 HDLCHOL, for HDLCHOL; P001 TOTCHOL, for TOTCHOL; and M021 SCHISTOc, for MSCHIST.
- SexM1F2 is a “dummy” variable that was coded with 1 assigned to males and 2 to females. As such, it essentially measures the “degree of femaleness” of the respondents. Being female is generally protective against cardiovascular disease, a situation that reverts itself a bit after menopause.
- MVASC is a composite measure of the two following variables, provided as component measures of mortality from all cardiovascular diseases: M058 ALLVASCb (ages 0-34), and M059 ALLVASCc (ages 35-69). A couple of obvious problems: (a) they does not include data on people older than 69; and (b) they seem to capture a lot of diseases, including some that do not seem like typical cardiovascular diseases. A factor analysis was conducted, and the loadings and cross-loadings suggested good validity. Composite reliability was also good. So essentially MVASC is measured here as a “latent variable” with two “indicators”. Why do this? The reason is that it reduces the biasing effects of incomplete data and measurement error (e.g., exclusion of folks older than 69). By the way, there is always some measurement error in any dataset.
- This note is related to measurement error in connection with the indicators for MVASC. There is something odd about the variables M058 ALLVASCb (ages 0-34), and M059 ALLVASCc (ages 35-69). According to the dataset, mortality from cardiovascular diseases for ages 0-34 is typically higher than for 35-69, for many counties. Given the good validity and reliability for MVASC as a latent variable, it is possible that the values for these two indicator variables were simply swapped by mistake.
Thursday, May 27, 2010
Postprandial glucose levels, HbA1c, and arterial stiffness: Compared to glucose, lipids are not even on the radar screen
Postprandial glucose levels are the levels of blood glucose after meals. In Western urban environments, the main contributors to elevated postprandial glucose are foods rich in refined carbohydrates and sugars. While postprandial glucose levels may vary somewhat erratically, they are particularly elevated in the morning after breakfast. The main reason for this is that breakfast, in Western urban environments, is typically very high in refined carbohydrates and sugars.
HbA1c, or glycated hemoglobin, is a measure of average blood glucose over a period of a few months. Blood glucose glycates (i.e., sticks to) hemoglobin, a protein found in red blood cells. Red blood cells are relatively long-lived, lasting approximately 3 months. Thus HbA1c (given in percentages) is a good indicator of average blood glucose levels, if you don’t suffer from anemia or a few other blood abnormalities.
Based on HbA1c, one can then estimate his or her average blood glucose level for the previous 3 months or so before the test, using one of the following equations, depending on whether the measurement is in mg/dl or mmol/l.
Average blood glucose (mg/dl) = 28.7 × HbA1c − 46.7
Average blood glucose (mmol/l) = 1.59 × HbA1c − 2.59
Elevated blood glucose levels cause damage in the body primarily through glycation, which leads to the formation of advanced glycation endproducts (AGEs). Given this, HbA1c can be seen as a proxy for the level of damage done by elevated blood glucose levels to various body tissues. This damage occurs over time; often after many years of high blood glucose levels. It includes kidney damage, neurological damage, cardiovascular damage, and damage to the retina.
Most regular blood exams focus on fasting blood glucose as a measure of glucose metabolism status. Many medical practitioners have as a target a fasting blood glucose level of 125 mg/dl (7 mmol/l) or less, and largely disregard postprandial glucose levels or HbA1c in their management of glucose metabolism. Leiter and colleagues (2005; full reference at the end of this post) showed that this focus on fasting blood glucose is a mistake. They are not alone; many others made this point, including some very knowledgeable bloggers who focus on diabetes (see “Interesting links” section of this blog). Leiter and colleagues (2005) also provided some interesting graphs and figures, including eye-opening correlations between various variables and arterial stiffness. The figure below (click to enlarge) shows the contribution of postprandial glucose to HbA1c.
Note that the lower the HbA1c is in the figure (horizontal axis), the higher is the postprandial glucose contribution to HbA1c. And, the lower the HbA1c, the closer the individuals are to what one could consider having a perfectly normal HbA1c level (around 5 percent). That is, only for individuals whose HbA1c levels are very high, fasting blood glucose levels are relatively reliable measures of the tissue damage done be elevated blood glucose levels.
The table below (click to enlarge) shows P values associated with the impact of various variables (listed on the leftmost column) on arterial stiffness. This measure, arterial stiffness, is strongly associated with an increased risk of cardiovascular events. Look at the middle column showing P values adjusted for age and height. The lower the P value, the more a variable affects arterial stiffness. The variable with the lowest P value by far is 2-hour postprandial blood glucose; the blood glucose levels measured 2 hours after meals.
Fasting glucose levels were reported to be statistically insignificant because of the P = 0.049, in terms of their effect on arterial stiffness, but this P value is actually significant, although barely, at the 0.05 level (95 percent confidence). Interestingly, the following measures are not even on the radar screen, as far as arterial stiffness is concerned: systolic blood pressure, LDL cholesterol, HDL cholesterol, triglycerides, and fasting insulin levels.
What about the lipid hypothesis, and the “bad” LDL cholesterol!? This study is telling us that these are not very relevant for arterial stiffness when we control for the effect of blood glucose measures. Not even fasting insulin levels matters much! Wait, not even HDL!!! A high HDL has been definitely shown to be protective, but when we look at the relative magnitude of various effects, the story is a bit different. A high HDL’s protective effect exists, but it is dwarfed by the negative effect of high blood glucose levels, especially after meals, in the context of cardiovascular disease.
What all this points at is what we could call a postprandial glucose hypothesis: Lower your postprandial glucose levels, and live a longer, healthier life! And, by the way, if your postprandial glucose levels are under control, lipids do not matter much! Or maybe your lipids will fall into place, without any need for statin drugs, after your postprandial glucose levels are under control. One way or another, the outcome will be a positive one. That is what the data from this study is telling us.
How do you lower your postprandial glucose levels?
A good way to start is to remove foods rich in refined carbohydrates and sugars from your diet. Almost all of these are foods engineered by humans with the goal of being addictive; they usually come in boxes and brightly colored plastic wraps. They are not hard to miss. They are typically in the central aisles of supermarkets. The sooner you remove them from your diet, the better. The more completely you do this, the better.
Note that the evidence discussed in this post is in connection with blood glucose levels, not glucose metabolism per se. If you have impaired glucose metabolism (e.g., diabetes type 2), you can still avoid a lot of problems if you effectively control your blood glucose levels. You may have to be a bit more aggressive, adding low carbohydrate dieting (as in the Atkins or Optimal diets) to the removal of refined carbohydrates and sugars from your diet; the latter is in many ways similar to adopting a Paleolithic diet. You may have to take some drugs, such as Metformin (a.k.a. Glucophage). But you are certainly not doomed if you are diabetic.
Reference:
Leiter, L.A., Ceriello, A., Davidson, J.A., Hanefeld, M., Monnier, L., Owens, D.R., Tajima, N., & Tuomilehto, J. (2005). Postprandial glucose regulation: New data and new implications. Clinical Therapeutics, 27(2), S42-S56.
HbA1c, or glycated hemoglobin, is a measure of average blood glucose over a period of a few months. Blood glucose glycates (i.e., sticks to) hemoglobin, a protein found in red blood cells. Red blood cells are relatively long-lived, lasting approximately 3 months. Thus HbA1c (given in percentages) is a good indicator of average blood glucose levels, if you don’t suffer from anemia or a few other blood abnormalities.
Based on HbA1c, one can then estimate his or her average blood glucose level for the previous 3 months or so before the test, using one of the following equations, depending on whether the measurement is in mg/dl or mmol/l.
Average blood glucose (mg/dl) = 28.7 × HbA1c − 46.7
Average blood glucose (mmol/l) = 1.59 × HbA1c − 2.59
Elevated blood glucose levels cause damage in the body primarily through glycation, which leads to the formation of advanced glycation endproducts (AGEs). Given this, HbA1c can be seen as a proxy for the level of damage done by elevated blood glucose levels to various body tissues. This damage occurs over time; often after many years of high blood glucose levels. It includes kidney damage, neurological damage, cardiovascular damage, and damage to the retina.
Most regular blood exams focus on fasting blood glucose as a measure of glucose metabolism status. Many medical practitioners have as a target a fasting blood glucose level of 125 mg/dl (7 mmol/l) or less, and largely disregard postprandial glucose levels or HbA1c in their management of glucose metabolism. Leiter and colleagues (2005; full reference at the end of this post) showed that this focus on fasting blood glucose is a mistake. They are not alone; many others made this point, including some very knowledgeable bloggers who focus on diabetes (see “Interesting links” section of this blog). Leiter and colleagues (2005) also provided some interesting graphs and figures, including eye-opening correlations between various variables and arterial stiffness. The figure below (click to enlarge) shows the contribution of postprandial glucose to HbA1c.
Note that the lower the HbA1c is in the figure (horizontal axis), the higher is the postprandial glucose contribution to HbA1c. And, the lower the HbA1c, the closer the individuals are to what one could consider having a perfectly normal HbA1c level (around 5 percent). That is, only for individuals whose HbA1c levels are very high, fasting blood glucose levels are relatively reliable measures of the tissue damage done be elevated blood glucose levels.
The table below (click to enlarge) shows P values associated with the impact of various variables (listed on the leftmost column) on arterial stiffness. This measure, arterial stiffness, is strongly associated with an increased risk of cardiovascular events. Look at the middle column showing P values adjusted for age and height. The lower the P value, the more a variable affects arterial stiffness. The variable with the lowest P value by far is 2-hour postprandial blood glucose; the blood glucose levels measured 2 hours after meals.
Fasting glucose levels were reported to be statistically insignificant because of the P = 0.049, in terms of their effect on arterial stiffness, but this P value is actually significant, although barely, at the 0.05 level (95 percent confidence). Interestingly, the following measures are not even on the radar screen, as far as arterial stiffness is concerned: systolic blood pressure, LDL cholesterol, HDL cholesterol, triglycerides, and fasting insulin levels.
What about the lipid hypothesis, and the “bad” LDL cholesterol!? This study is telling us that these are not very relevant for arterial stiffness when we control for the effect of blood glucose measures. Not even fasting insulin levels matters much! Wait, not even HDL!!! A high HDL has been definitely shown to be protective, but when we look at the relative magnitude of various effects, the story is a bit different. A high HDL’s protective effect exists, but it is dwarfed by the negative effect of high blood glucose levels, especially after meals, in the context of cardiovascular disease.
What all this points at is what we could call a postprandial glucose hypothesis: Lower your postprandial glucose levels, and live a longer, healthier life! And, by the way, if your postprandial glucose levels are under control, lipids do not matter much! Or maybe your lipids will fall into place, without any need for statin drugs, after your postprandial glucose levels are under control. One way or another, the outcome will be a positive one. That is what the data from this study is telling us.
How do you lower your postprandial glucose levels?
A good way to start is to remove foods rich in refined carbohydrates and sugars from your diet. Almost all of these are foods engineered by humans with the goal of being addictive; they usually come in boxes and brightly colored plastic wraps. They are not hard to miss. They are typically in the central aisles of supermarkets. The sooner you remove them from your diet, the better. The more completely you do this, the better.
Note that the evidence discussed in this post is in connection with blood glucose levels, not glucose metabolism per se. If you have impaired glucose metabolism (e.g., diabetes type 2), you can still avoid a lot of problems if you effectively control your blood glucose levels. You may have to be a bit more aggressive, adding low carbohydrate dieting (as in the Atkins or Optimal diets) to the removal of refined carbohydrates and sugars from your diet; the latter is in many ways similar to adopting a Paleolithic diet. You may have to take some drugs, such as Metformin (a.k.a. Glucophage). But you are certainly not doomed if you are diabetic.
Reference:
Leiter, L.A., Ceriello, A., Davidson, J.A., Hanefeld, M., Monnier, L., Owens, D.R., Tajima, N., & Tuomilehto, J. (2005). Postprandial glucose regulation: New data and new implications. Clinical Therapeutics, 27(2), S42-S56.
Friday, May 7, 2010
Niacin and its effects on growth hormone, glucagon, cortisol, blood lipids, mental disorders, and fasting glucose levels
Niacin is a very interesting vitamin. It is also known as vitamin B3, or nicotinic acid. It is an essential vitamin whose deficiency leads to a dreadful disease known as pellagra. In large doses of 1 to 3 g per day it has several effects on blood lipids, including these: it increases HDL cholesterol, decreases triglycerides, and decreases Lp(a). Given that this is essentially a reversal of the metabolic syndrome, for those who are on their way to developing it, niacin must really do something good for our body. Niacin is also a powerful antioxidant.
The lipid modification effects of niacin are so consistent across a broad spectrum of the population that some companies that commercialize niacin-based products guarantee some measure of those effects. The graphs below (click to enlarge) are from Arizona Pharmaceuticals, a company that commercializes an instant-release niacin formulation called Nialor (see: arizonapharmaceuticals.com). The graphs show the peak effects on HDL cholesterol and triglycerides at the recommended dose, which is 1.5 g per day. The company guarantees effects; not the peak effects shown, but effects that are large enough to have clinical significance.
Niacin also has been used in the treatment of various mental disorders, including schizophrenia. Its effectiveness in this domain (mental disease) is still under debate. Yet many people, including reputable mental health researchers, swear by it. Empirical research suggests beyond much doubt that niacin helps in the treatment of depression and bipolar disorder.
Abram Hoffer, a Canadian psychiatrist who died in 2009, at the age of 91, has discussed at length the many beneficial health effects of niacin. He was also a niacin user. He argued that it can even make people live longer, and be generally healthier and more active. The effect on longevity may sound far-fetched, but there is empirical data supporting this hypothesis as well. (For more, see this book.)
By the way, moderate niacin supplementation seems to increase the milk output of cows, without any effect on milk composition.
Most people dislike the sensation that is caused by niacin, the “niacin flush”. This is a temporary sensation similar to that of sunburn covering one’s full torso and face. It goes away after a few minutes. This is niacin’s main undesirable side effect at doses up to 3 g per day. Higher doses are not recommended, and can be toxic to the liver.
Nobody seems to understand very well how niacin works. This leads to some confusion. Many people think that niacin inhibits the production of VLDL, free fatty acids, and ketones; preventing the use of fat as an energy source. And it does!
So it makes you fat, right?
No, because these effects are temporary, and are followed, often after 3 to 5 hours, by a large increase in circulating growth hormone, cortisol and glucagon. These hormones are associated with (maybe they cause, maybe are caused by) a large increase in free fatty acids and ketones in circulation, but not with an increase in VLDL secretion by the liver. So ketosis is at first inhibited by niacin, and then comes in full force after a few hours.
The decreased VLDL secretion is no surprise, because VLDL is not really needed in large quantities if muscle tissues (including the heart) are being fed what they really like: free fatty acids and ketones. When VLDL particles are secreted by the liver in small numbers, they tend to be large. As they shrink in size after delivering their lipid content to muscle tissues, they become large LDL particles; too large to cross the endothelial gaps and cause plaque formation.
It is as if niacin held you back for a few hours, in terms of fat burning, and then released you with a strong push.
Since niacin does not seem to suppress the secretion of chylomicrons by the intestines, it should be taken with meals. The meals do not necessarily have to have any carbohydrates in them. If you take niacin while fasting, you may feel “funny” and somewhat weak, because of the decrease in VLDL, free fatty acids, and ketones in circulation. These, particularly the free fatty acids and ketones, are important sources of energy in the fasted state.
Given niacin’s delayed effects, it does not seem to make much sense to take slow release niacin of any kind. In fact, the form of niacin that seems to work best is the instant-release one, the one that gives you the flush. It may be a good idea to wait until 3 to 5 hours after you take it to do heavy exercise. You may feel a surge of energy 3 to 5 hours after taking it, when the delayed effects kick in.
The delayed effects of niacin on growth hormone, cortisol and glucagon are probably the reasons why people taking niacin frequently see a small increase in fasting glucose levels. This increase is usually of a few percentage points, but can be a bit higher in some people. Growth hormone, cortisol and particularly glucagon increase blood glucose levels; and the blood levels of these hormones naturally rise in the morning to get you ready for the day ahead. Niacin seems to boost that. Hence the increase in fasting blood glucose levels. This appears to be a benign effect, easily counterbalanced by niacin’s many benefits.
In spite of a possible increase in fasting glucose levels, there is no evidence that niacin increases average blood glucose levels. If it did, that would not be a good thing. In fact, it has been argued that niacin intake can be part of an effective approach to treating diabetes; Robert C. Atkins discussed this in his Vita-Nutrient Solution book.
Niacin’s effects on lipids are somewhat similar to those of low carbohydrate dieting. For example, both lead to a decrease in fasting triglycerides and an increase in HDL cholesterol. But the mechanisms by which those effects are achieved appear to be rather different.
References:
Quabbe, H.J., Trompke, M., & Luyckx, A.S. (1983). Influence of ketone body infusion on plasma growth hormone and glucagon in man. J. Clin Endocrinol Metab., 57(3):613-8.
Quabbe, H.J., Luyckx, A.S., L'age M., & Schwarz, C. (1983). Growth hormone, cortisol, and glucagon concentrations during plasma free fatty acid depression: different effects of nicotinic acid and an adenosine derivative (BM 11.189). J. Clin Endocrinol Metab., 57(2):410-4.
Schade, D.S., Woodside, W., & Eaton, R.P. (1979). The role of glucagon in the regulation of plasma lipids. Metabolism, 28(8):874-86.
The lipid modification effects of niacin are so consistent across a broad spectrum of the population that some companies that commercialize niacin-based products guarantee some measure of those effects. The graphs below (click to enlarge) are from Arizona Pharmaceuticals, a company that commercializes an instant-release niacin formulation called Nialor (see: arizonapharmaceuticals.com). The graphs show the peak effects on HDL cholesterol and triglycerides at the recommended dose, which is 1.5 g per day. The company guarantees effects; not the peak effects shown, but effects that are large enough to have clinical significance.
Niacin also has been used in the treatment of various mental disorders, including schizophrenia. Its effectiveness in this domain (mental disease) is still under debate. Yet many people, including reputable mental health researchers, swear by it. Empirical research suggests beyond much doubt that niacin helps in the treatment of depression and bipolar disorder.
Abram Hoffer, a Canadian psychiatrist who died in 2009, at the age of 91, has discussed at length the many beneficial health effects of niacin. He was also a niacin user. He argued that it can even make people live longer, and be generally healthier and more active. The effect on longevity may sound far-fetched, but there is empirical data supporting this hypothesis as well. (For more, see this book.)
By the way, moderate niacin supplementation seems to increase the milk output of cows, without any effect on milk composition.
Most people dislike the sensation that is caused by niacin, the “niacin flush”. This is a temporary sensation similar to that of sunburn covering one’s full torso and face. It goes away after a few minutes. This is niacin’s main undesirable side effect at doses up to 3 g per day. Higher doses are not recommended, and can be toxic to the liver.
Nobody seems to understand very well how niacin works. This leads to some confusion. Many people think that niacin inhibits the production of VLDL, free fatty acids, and ketones; preventing the use of fat as an energy source. And it does!
So it makes you fat, right?
No, because these effects are temporary, and are followed, often after 3 to 5 hours, by a large increase in circulating growth hormone, cortisol and glucagon. These hormones are associated with (maybe they cause, maybe are caused by) a large increase in free fatty acids and ketones in circulation, but not with an increase in VLDL secretion by the liver. So ketosis is at first inhibited by niacin, and then comes in full force after a few hours.
The decreased VLDL secretion is no surprise, because VLDL is not really needed in large quantities if muscle tissues (including the heart) are being fed what they really like: free fatty acids and ketones. When VLDL particles are secreted by the liver in small numbers, they tend to be large. As they shrink in size after delivering their lipid content to muscle tissues, they become large LDL particles; too large to cross the endothelial gaps and cause plaque formation.
It is as if niacin held you back for a few hours, in terms of fat burning, and then released you with a strong push.
Since niacin does not seem to suppress the secretion of chylomicrons by the intestines, it should be taken with meals. The meals do not necessarily have to have any carbohydrates in them. If you take niacin while fasting, you may feel “funny” and somewhat weak, because of the decrease in VLDL, free fatty acids, and ketones in circulation. These, particularly the free fatty acids and ketones, are important sources of energy in the fasted state.
Given niacin’s delayed effects, it does not seem to make much sense to take slow release niacin of any kind. In fact, the form of niacin that seems to work best is the instant-release one, the one that gives you the flush. It may be a good idea to wait until 3 to 5 hours after you take it to do heavy exercise. You may feel a surge of energy 3 to 5 hours after taking it, when the delayed effects kick in.
The delayed effects of niacin on growth hormone, cortisol and glucagon are probably the reasons why people taking niacin frequently see a small increase in fasting glucose levels. This increase is usually of a few percentage points, but can be a bit higher in some people. Growth hormone, cortisol and particularly glucagon increase blood glucose levels; and the blood levels of these hormones naturally rise in the morning to get you ready for the day ahead. Niacin seems to boost that. Hence the increase in fasting blood glucose levels. This appears to be a benign effect, easily counterbalanced by niacin’s many benefits.
In spite of a possible increase in fasting glucose levels, there is no evidence that niacin increases average blood glucose levels. If it did, that would not be a good thing. In fact, it has been argued that niacin intake can be part of an effective approach to treating diabetes; Robert C. Atkins discussed this in his Vita-Nutrient Solution book.
Niacin’s effects on lipids are somewhat similar to those of low carbohydrate dieting. For example, both lead to a decrease in fasting triglycerides and an increase in HDL cholesterol. But the mechanisms by which those effects are achieved appear to be rather different.
References:
Quabbe, H.J., Trompke, M., & Luyckx, A.S. (1983). Influence of ketone body infusion on plasma growth hormone and glucagon in man. J. Clin Endocrinol Metab., 57(3):613-8.
Quabbe, H.J., Luyckx, A.S., L'age M., & Schwarz, C. (1983). Growth hormone, cortisol, and glucagon concentrations during plasma free fatty acid depression: different effects of nicotinic acid and an adenosine derivative (BM 11.189). J. Clin Endocrinol Metab., 57(2):410-4.
Schade, D.S., Woodside, W., & Eaton, R.P. (1979). The role of glucagon in the regulation of plasma lipids. Metabolism, 28(8):874-86.
Sunday, April 11, 2010
The Friedewald and Iranian equations: Fasting triglycerides can seriously distort calculated LDL
Standard lipid profiles provide LDL cholesterol measures based on equations that usually have the following as their inputs (or independent variables): total cholesterol, HDL cholesterol, and triglycerides.
Yes, LDL cholesterol is not measured directly in standard lipid profile tests! This is indeed surprising, since cholesterol-lowering drugs with negative side effects are usually prescribed based on estimated (or "fictitious") LDL cholesterol levels.
The most common of these equations is the Friedewald equation. Through the Friedewald equation, LDL cholesterol is calculated as follows (where TC = total cholesterol, and TG = triglycerides). The inputs and result are in mg/dl.
LDL = TC – HDL – TG / 5
Here is one of the problems with the Friedewald equation. Let us assume that an individual has the following lipid profile numbers: TC = 200, HDL = 50, and trigs. = 150. The calculated LDL will be 120. Let us assume that this same individual reduces triglycerides to 50, from the previous 150, keeping all of the other measures constant. This is a major improvement. However, the calculated LDL will now be 140, and a doctor will tell this person to consider taking statins!
There is evidence that, for individuals with low fasting triglycerides, a more precise equation is one that has come to be known as the “Iranian equation”. The equation has been proposed by Iranian researchers in an article published in the Archives of Iranian Medicine (Ahmadi et al., 2008), hence its nickname. Through the Iranian equation, LDL is calculated as follows. Again, the inputs and result are in mg/dl.
LDL = TC / 1.19 + TG / 1.9 – HDL / 1.1 – 38
The Iranian equation is based on linear regression modeling, which is a good sign, although I would have liked it even better if it was based on nonlinear regression modeling. The reason is that relationships between variables describing health-related phenomena are often nonlinear, leading to biased linear estimations. With a good nonlinear analysis algorithm, a linear relationship will also be captured; that is, the “curve” that describes the relationship will default to a line if the relationship is truly linear (see: warppls.com).
Anyway, an online calculator that implements both equations (Friedewald and Iranian) is linked here; it was the top Google hit on a search for “Iranian equation LDL” at the time of this post’s writing.
As you will see if you try it, the online calculator linked above is useful in showing the difference in calculated LDL cholesterol, using both equations, when fasting triglycerides are very low (e.g., below 50).
The Iranian equation yields high values of LDL cholesterol when triglycerides are high; much higher than those generated by the Friedewald equation. If those are not overestimations (and there is some evidence that, if they are, it is not by much), they describe an alarming metabolic pattern, because high triglycerides are associated with small-dense LDL particles. These particles are the most potentially atherogenic of the LDL particles, in the presence of other factors such as chronic inflammation.
In other words, the Iranian equation gives a clearer idea than the Friedewald equation about the negative health effects of high triglycerides. You need a large number of small-dense LDL particles to carry a high amount of LDL cholesterol.
An even more precise measure of LDL particle configuration is the VAP test; this post has a link to a PDF file with a sample VAP test report.
Reference:
Ahmadi SA, Boroumand MA, Gohari-Moghaddam K, Tajik P, Dibaj SM. (2008). The impact of low serum triglyceride on LDL-cholesterol estimation. Archives of Iranian Medicine, 11(3), 318-21.
Yes, LDL cholesterol is not measured directly in standard lipid profile tests! This is indeed surprising, since cholesterol-lowering drugs with negative side effects are usually prescribed based on estimated (or "fictitious") LDL cholesterol levels.
The most common of these equations is the Friedewald equation. Through the Friedewald equation, LDL cholesterol is calculated as follows (where TC = total cholesterol, and TG = triglycerides). The inputs and result are in mg/dl.
LDL = TC – HDL – TG / 5
Here is one of the problems with the Friedewald equation. Let us assume that an individual has the following lipid profile numbers: TC = 200, HDL = 50, and trigs. = 150. The calculated LDL will be 120. Let us assume that this same individual reduces triglycerides to 50, from the previous 150, keeping all of the other measures constant. This is a major improvement. However, the calculated LDL will now be 140, and a doctor will tell this person to consider taking statins!
There is evidence that, for individuals with low fasting triglycerides, a more precise equation is one that has come to be known as the “Iranian equation”. The equation has been proposed by Iranian researchers in an article published in the Archives of Iranian Medicine (Ahmadi et al., 2008), hence its nickname. Through the Iranian equation, LDL is calculated as follows. Again, the inputs and result are in mg/dl.
LDL = TC / 1.19 + TG / 1.9 – HDL / 1.1 – 38
The Iranian equation is based on linear regression modeling, which is a good sign, although I would have liked it even better if it was based on nonlinear regression modeling. The reason is that relationships between variables describing health-related phenomena are often nonlinear, leading to biased linear estimations. With a good nonlinear analysis algorithm, a linear relationship will also be captured; that is, the “curve” that describes the relationship will default to a line if the relationship is truly linear (see: warppls.com).
Anyway, an online calculator that implements both equations (Friedewald and Iranian) is linked here; it was the top Google hit on a search for “Iranian equation LDL” at the time of this post’s writing.
As you will see if you try it, the online calculator linked above is useful in showing the difference in calculated LDL cholesterol, using both equations, when fasting triglycerides are very low (e.g., below 50).
The Iranian equation yields high values of LDL cholesterol when triglycerides are high; much higher than those generated by the Friedewald equation. If those are not overestimations (and there is some evidence that, if they are, it is not by much), they describe an alarming metabolic pattern, because high triglycerides are associated with small-dense LDL particles. These particles are the most potentially atherogenic of the LDL particles, in the presence of other factors such as chronic inflammation.
In other words, the Iranian equation gives a clearer idea than the Friedewald equation about the negative health effects of high triglycerides. You need a large number of small-dense LDL particles to carry a high amount of LDL cholesterol.
An even more precise measure of LDL particle configuration is the VAP test; this post has a link to a PDF file with a sample VAP test report.
Reference:
Ahmadi SA, Boroumand MA, Gohari-Moghaddam K, Tajik P, Dibaj SM. (2008). The impact of low serum triglyceride on LDL-cholesterol estimation. Archives of Iranian Medicine, 11(3), 318-21.
Tuesday, April 6, 2010
Low fasting triglycerides: A marker for large-buoyant LDL particles
Small-dense LDL particles are particles that are significantly smaller than the gaps in the endothelium. The endothelium is a thin layer of cells that line the interior of arteries. Those gaps are about 25-26 nanometers (nm) in diameter. Small-dense LDL particles can contribute a lot more to the formation of atheromas (atherosclerotic plaques) in predisposed individuals than large-buoyant LDL particles.
Note that typically LDL particles are about 23-25 nm in diameter in most people, and yet not everybody develops atheromas. It is illogical to believe that evolution made LDL particles within those ranges of size to harm us, given the size of the gaps in the endothelium, unless you believe in something like this joke theory. There are underlying factors that make individuals much more prone to the development of atheromas than others.
One of those factors is chronic inflammation, which is caused by: chronic stress, excessive exercise (aerobic or anaerobic), and a diet rich in refined carbohydrates (e.g., white bread, pasta) and refined sugars (e.g., high fructose corn syrup, table sugar).
Can a standard lipid profile report tell me anything about my LDL particle pattern?
Yes, check you fasting triglycerides. If they are below 70 mg/dL, it is very likely that you have a predominance of large-buoyant LDL particles in your blood. That is, your LDL particle pattern is most likely Pattern A (see figure below, from: www.degomamd.com), the least atherogenic of the patterns identified by a Vertical Auto Profile (VAP) test. This test is more sophisticated than a standard lipid profile test, where the LDL cholesterol is typically calculated. For a sample VAP test report, see this PDF file from Atherotech.
So, you can get a rough idea about your LDL pattern type only by checking your fasting triglyceride levels on a standard lipid profile test report, if you cannot or do not want to have a VAP test done. The higher your fasting triglyceride levels are, above 70, the more likely it is that your LDL particle pattern is Pattern B, which is the most potentially atherogenic pattern.
Large-buoyant LDL particles often lead to high measured LDL cholesterol levels. This situation is analogous to that of water-filled balloons. If you have 10 balloons, each holding 0.5 L of water, then your total water amount is 5 L. If the same balloons are filled with 1 L of water each, then your total water amount is 10 L. That is, even though the number of LDL particles (analogous to the number of balloons) may be the same as that of a person with low LDL cholesterol, large-buoyant LDL particles have more cholesterol (water content in each balloon) in them, and lead to higher measured LDL cholesterol (total amount of water in the balloons) levels.
This leads to the counterintuitive situation where your LDL cholesterol levels go up, and your risk of developing cardiovascular disease actually goes down.
Also worth keeping in mind is that fasting triglyceride levels are strongly and negatively correlated with HDL cholesterol levels. The higher your fasting triglyceride levels are, usually the lower are your HDL cholesterol levels. The latter are also provided in standard lipid profile reports.
How do you decrease your fasting triglycerides?
A good way to start is to do some of the things that increase your HDL cholesterol.
References:
Elliott, W.H., & Elliott, D.C. (2009). Biochemistry and molecular biology. 4th Edition. New York: NY: Oxford University Press.
Lemanski, P.E. (2004). Beyond routine cholesterol testing: The role of LDL particle size assessment. CDPHP Medical Messenger, May 2004.
Note that typically LDL particles are about 23-25 nm in diameter in most people, and yet not everybody develops atheromas. It is illogical to believe that evolution made LDL particles within those ranges of size to harm us, given the size of the gaps in the endothelium, unless you believe in something like this joke theory. There are underlying factors that make individuals much more prone to the development of atheromas than others.
One of those factors is chronic inflammation, which is caused by: chronic stress, excessive exercise (aerobic or anaerobic), and a diet rich in refined carbohydrates (e.g., white bread, pasta) and refined sugars (e.g., high fructose corn syrup, table sugar).
Can a standard lipid profile report tell me anything about my LDL particle pattern?
Yes, check you fasting triglycerides. If they are below 70 mg/dL, it is very likely that you have a predominance of large-buoyant LDL particles in your blood. That is, your LDL particle pattern is most likely Pattern A (see figure below, from: www.degomamd.com), the least atherogenic of the patterns identified by a Vertical Auto Profile (VAP) test. This test is more sophisticated than a standard lipid profile test, where the LDL cholesterol is typically calculated. For a sample VAP test report, see this PDF file from Atherotech.
So, you can get a rough idea about your LDL pattern type only by checking your fasting triglyceride levels on a standard lipid profile test report, if you cannot or do not want to have a VAP test done. The higher your fasting triglyceride levels are, above 70, the more likely it is that your LDL particle pattern is Pattern B, which is the most potentially atherogenic pattern.
Large-buoyant LDL particles often lead to high measured LDL cholesterol levels. This situation is analogous to that of water-filled balloons. If you have 10 balloons, each holding 0.5 L of water, then your total water amount is 5 L. If the same balloons are filled with 1 L of water each, then your total water amount is 10 L. That is, even though the number of LDL particles (analogous to the number of balloons) may be the same as that of a person with low LDL cholesterol, large-buoyant LDL particles have more cholesterol (water content in each balloon) in them, and lead to higher measured LDL cholesterol (total amount of water in the balloons) levels.
This leads to the counterintuitive situation where your LDL cholesterol levels go up, and your risk of developing cardiovascular disease actually goes down.
Also worth keeping in mind is that fasting triglyceride levels are strongly and negatively correlated with HDL cholesterol levels. The higher your fasting triglyceride levels are, usually the lower are your HDL cholesterol levels. The latter are also provided in standard lipid profile reports.
How do you decrease your fasting triglycerides?
A good way to start is to do some of the things that increase your HDL cholesterol.
References:
Elliott, W.H., & Elliott, D.C. (2009). Biochemistry and molecular biology. 4th Edition. New York: NY: Oxford University Press.
Lemanski, P.E. (2004). Beyond routine cholesterol testing: The role of LDL particle size assessment. CDPHP Medical Messenger, May 2004.
Sunday, March 28, 2010
LDL, chylomicrons, HDL, and atherosclerosis: A lazy Sunday theory
Notes:
- This post is a joke, admittedly a weird one, which is why it is labeled “humor” and is filed under “Abstract humor”.
- I apologize for this spoiler. Some people probably like humor posts better if they do not know what they are in advance, but several others may think that reading a post like this is a waste of their time. If you are in the latter category, move on to another post! If not, here it goes …
Today I was spending some time under the sun, in one of the year’s 364 sunny days in Laredo, Texas. The goal was to see if I could obtain a precise count of the number of advanced glycation endproducts (a.k.a. AGEs) that would form as my skin was exposed to the sun’s damaging rays.
Then I read a post by Peter at Hyperlipid, and inspiration consumed me. A new theory was born regarding the interplay of LDL, chylomicrons, HDL, and atherosclerosis. By the way, Peter is a fat genius, by which I mean a genius regarding all fat issues – who happens to be thin.
A key observation forms the main pillar on which this new theory solidly rests:
The endothelium gaps, which let atherogenic particles enter into the forbidden area and do their damage, are around 25 nanometers in diameter. And what is the typical size of LDL particles? You guessed it, 25 nanometers in diameter! And guess what more, quite a few of the chylomicrons, another group of particles that would elicit immediate revulsion in any normal human being, are even smaller than 25 nanometers in diameter; those atherogenic pests!
So here is the theory, in a nutshell. A 500-page book will clearly be needed to discuss it in more detail.
The Devil created LDL particles to kill us all. But LDL particles were not such effective killers, because the Devil, trying to pack as much killer cholesterol into them, ended up making them too big! At 25 nanometers in diameter, on average, they basically had to squeeze their way into the forbidden area.
Since LDL particles were not doing a good enough job, the Devil also created chylomicrons, and those chaotic pests come in all sizes. In fact, it is well known that the word chylomicron has a Greek origin: chylo = killer, micron = particle (Deth & Disis, 1999; full reference at the end of this post).
And, needless to say, LDL particles and chylomicrons are fat particles that make the blood kind of taste and smell like butter, a toxic substance often fed to laboratory rats and known for its powerful carcinogenic properties among all living creatures except descendants of Vlad the Impaler. The latter has long been rumored to have been one of the Devil's best buddies, so no surprise there.
Michael the Archangel, who dislikes the Devil, and usually takes a hands-on approach to dealing with those he dislikes, the Devil in particular, gave us HDL particles. If you have any doubts about Michael’s hands-on approach, check the picture below (from: Wikipedia), which clearly shows what Michael had already done to the Devil. And that was over a relatively minor disagreement.
And don’t think about trying to discredit this theory by asking why HDL particles are so small compared with LDL particles and chylomicrons! This is easy. For the same reason that David was small and Goliath big!
But those nasty particles, the LDLs and chylomicrons, weren't only two big bullies, they were two against one. HDL particles were doing a valiant job at fighting the damage done by the Devil’s two evil particles, but not quite enough to save everybody from atherosclerosis.
Michael cried foul, and threatened to give the Devil another lesson. God, seeing this, said: Michael, no, mankind must be given a choice! If men and women want to gorge on the fatty flesh of the beasts they savagely slaughter, let them sin and face the consequences.
And so it was.
This theory probably needs some adjustments and refinements based on analysis of refereed research, especially solid research supported by drug manufacturers, and consultation with the most interesting man in the world. But I am pretty confident it can, after adjustments and refinements, pass the test of time.
The only nagging problem is the Original Sin. To the best of my knowledge, it was not eating the fatty flesh of beasts. It was eating a very sweet apple …
Reference:
Deth, R., & Disis, M. (1999, Feb 31). The origins of killer lipids: An evolutionary-theological perspective. The Lipid Review, 123(7), 77-66.
- This post is a joke, admittedly a weird one, which is why it is labeled “humor” and is filed under “Abstract humor”.
- I apologize for this spoiler. Some people probably like humor posts better if they do not know what they are in advance, but several others may think that reading a post like this is a waste of their time. If you are in the latter category, move on to another post! If not, here it goes …
***
Today I was spending some time under the sun, in one of the year’s 364 sunny days in Laredo, Texas. The goal was to see if I could obtain a precise count of the number of advanced glycation endproducts (a.k.a. AGEs) that would form as my skin was exposed to the sun’s damaging rays.
Then I read a post by Peter at Hyperlipid, and inspiration consumed me. A new theory was born regarding the interplay of LDL, chylomicrons, HDL, and atherosclerosis. By the way, Peter is a fat genius, by which I mean a genius regarding all fat issues – who happens to be thin.
A key observation forms the main pillar on which this new theory solidly rests:
The endothelium gaps, which let atherogenic particles enter into the forbidden area and do their damage, are around 25 nanometers in diameter. And what is the typical size of LDL particles? You guessed it, 25 nanometers in diameter! And guess what more, quite a few of the chylomicrons, another group of particles that would elicit immediate revulsion in any normal human being, are even smaller than 25 nanometers in diameter; those atherogenic pests!
So here is the theory, in a nutshell. A 500-page book will clearly be needed to discuss it in more detail.
The Devil created LDL particles to kill us all. But LDL particles were not such effective killers, because the Devil, trying to pack as much killer cholesterol into them, ended up making them too big! At 25 nanometers in diameter, on average, they basically had to squeeze their way into the forbidden area.
Since LDL particles were not doing a good enough job, the Devil also created chylomicrons, and those chaotic pests come in all sizes. In fact, it is well known that the word chylomicron has a Greek origin: chylo = killer, micron = particle (Deth & Disis, 1999; full reference at the end of this post).
And, needless to say, LDL particles and chylomicrons are fat particles that make the blood kind of taste and smell like butter, a toxic substance often fed to laboratory rats and known for its powerful carcinogenic properties among all living creatures except descendants of Vlad the Impaler. The latter has long been rumored to have been one of the Devil's best buddies, so no surprise there.
Michael the Archangel, who dislikes the Devil, and usually takes a hands-on approach to dealing with those he dislikes, the Devil in particular, gave us HDL particles. If you have any doubts about Michael’s hands-on approach, check the picture below (from: Wikipedia), which clearly shows what Michael had already done to the Devil. And that was over a relatively minor disagreement.
And don’t think about trying to discredit this theory by asking why HDL particles are so small compared with LDL particles and chylomicrons! This is easy. For the same reason that David was small and Goliath big!
But those nasty particles, the LDLs and chylomicrons, weren't only two big bullies, they were two against one. HDL particles were doing a valiant job at fighting the damage done by the Devil’s two evil particles, but not quite enough to save everybody from atherosclerosis.
Michael cried foul, and threatened to give the Devil another lesson. God, seeing this, said: Michael, no, mankind must be given a choice! If men and women want to gorge on the fatty flesh of the beasts they savagely slaughter, let them sin and face the consequences.
And so it was.
This theory probably needs some adjustments and refinements based on analysis of refereed research, especially solid research supported by drug manufacturers, and consultation with the most interesting man in the world. But I am pretty confident it can, after adjustments and refinements, pass the test of time.
The only nagging problem is the Original Sin. To the best of my knowledge, it was not eating the fatty flesh of beasts. It was eating a very sweet apple …
Reference:
Deth, R., & Disis, M. (1999, Feb 31). The origins of killer lipids: An evolutionary-theological perspective. The Lipid Review, 123(7), 77-66.
Friday, March 19, 2010
Online calculators to assess cardiovascular disease risk: No LDL needed
Researchers can build mathematical equations (sometimes referred to as structural equations) that predict health outcomes based on health factors. Those mathematical equations can then be used in online calculators.
This link takes you to a government-sponsored calculator based on the Framingham Heart Study. It estimates 10-year risk for “hard” coronary heart disease outcomes (myocardial infarction and coronary death).
As you will notice, the link above does not take family history of disease into consideration. A different risk calculator, linked here, estimates a risk score called the Reynold Risk Score. It takes hsCRP (high sensitivity C-reactive protein, an inflammation marker) and family history in addition to the Framingham parameters in its risk score calculation.
Neither calculator asks for LDL cholesterol levels. I wonder why.
This link takes you to a government-sponsored calculator based on the Framingham Heart Study. It estimates 10-year risk for “hard” coronary heart disease outcomes (myocardial infarction and coronary death).
As you will notice, the link above does not take family history of disease into consideration. A different risk calculator, linked here, estimates a risk score called the Reynold Risk Score. It takes hsCRP (high sensitivity C-reactive protein, an inflammation marker) and family history in addition to the Framingham parameters in its risk score calculation.
Neither calculator asks for LDL cholesterol levels. I wonder why.
Saturday, February 20, 2010
What should be my HDL cholesterol?
HDL cholesterol levels are a rough measure of HDL particle quantity in the blood. They actually tell us next to nothing about HDL particle type, although HDL cholesterol increases are usually associated with increases in LDL particle size. This a good thing, since small-dense LDL particles are associated with increased cardiovascular disease.
Most blood lipid panels reviewed by family doctors with patients give information about HDL status through measures of HDL cholesterol, provided in one of the standard units (e.g., mg/dl).
Study after study shows that HDL cholesterol levels, although imprecise, are a much better predictor of cardiovascular disease than LDL or total cholesterol levels. How high should be one’s HDL cholesterol? The answer to this question is somewhat dependent on each individual’s health profile, but most data suggest that a level greater than 60 mg/dl (1.55 mmol/l) is close to optimal for most people.
The figure below (from Eckardstein, 2008; full reference at the end of this post) plots incidence of coronary events in men (on the vertical axis), over a period of 10 years, against HDL cholesterol levels (on the horizontal axis). Note: IFG = impaired fasting glucose. This relationship is similar for women, particularly post-menopausal women. Pre-menopausal women usually have higher HDL cholesterol levels than men, and a low incidence of coronary events.
From the figure above, one can say that a diabetic man with about 55 mg/dl of HDL cholesterol will have approximately the same chance, on average, of having a coronary event (a heart attack) as a man with no risk factors and about 20 mg/dl of HDL cholesterol. That chance will be about 7 percent. With 20 mg/dl of HDL cholesterol, the chance of a diabetic man having a coronary event would approach 50 percent.
We can also conclude from the figure above that a man with no risk factors will have a 5 percent chance of having a coronary event if his HDL cholesterol is about 25 mg/dl; and about 2 percent if his HDL cholesterol is greater than 60 mg/dl. This a 60 percent reduction in risk, a risk that was low to start with because of the absence of risk factors.
HDL cholesterol levels greater than 60 are associated with significantly reduced risks of coronary events, particularly for those with diabetes (the graph does not take diabetes type into consideration). Much higher levels of HDL cholesterol (beyond 60) do not seem to be associated with much lower risk of coronary events.
Conversely, a very low HDL cholesterol level (below 25) is a major risk factor when other risk factors are also present, particularly: diabetes, hypertension (high blood pressure), and familial hypercholesteromia (gene-induced very elevated LDL cholesterol).
It is not yet clear whether HDL cholesterol is a cause of reduced cardiovascular disease, or just a marker of other health factors that lead to reduced risk for cardiovascular disease. Much of the empirical evidence suggests a causal relationship, and if this is the case then it may be a good idea to try to increase HDL levels. Even if HDL cholesterol is just a marker, the same strategy that increases it may also have a positive impact on the real causative factor of which HDL cholesterol is a marker.
What can one do to increase his or her HDL cholesterol? One way is to replace refined carbs and sugars with saturated fat and cholesterol in one’s diet. (I know that this sounds counterintuitive, but seems to work.) Another is to increase one’s vitamin D status, through sun exposure or supplementation.
Other therapeutic interventions can also be used to increase HDL; some more natural than others. The figure below (also from Eckardstein, 2008) shows the maximum effects of several therapeutic interventions to increase HDL cholesterol.
Among the therapeutic interventions shown in the figure above, taking nicotinic acid (niacin) in pharmacological doses, of 1 to 3 g per day (higher dosages may be toxic), is by far the most effective way of increasing one’s HDL cholesterol. Only the niacin that causes flush is effective in this respect. No-flush niacin preparations may have some anti-inflammatory effects, but do not cause increases in HDL cholesterol.
Rimonabant, which is second to niacin in its effect on HDL cholesterol, is an appetite suppressor that has been associated with serious side effects and, to be best of my knowledge, has been largely banned from use in pharmaceutical drugs.
Third in terms of effectiveness, among the factors shown in the figure, is moderate alcohol consumption. Running about 19 miles per week (2.7 miles per day) and taking fibrates are tied in forth place.
Many people think that they are having a major allergic reaction, and have a panic attack, when they experience the niacin flush. This usually happens several minutes after taking niacin, and depends on the dose and whether niacin was consumed with food or not. It is not uncommon for one’s entire torso to turn hot red, as though the person had had major sunburn. This reaction is harmless, and usually disappears after several minutes.
One could say that, with niacin: no “pain” (i.e., flush), no gain.
Reference:
von Eckardstein, A. (2008). HDL – a difficult friend. Drug Discovery Today: Disease Mechanisms, 5(3), 315-324.
Most blood lipid panels reviewed by family doctors with patients give information about HDL status through measures of HDL cholesterol, provided in one of the standard units (e.g., mg/dl).
Study after study shows that HDL cholesterol levels, although imprecise, are a much better predictor of cardiovascular disease than LDL or total cholesterol levels. How high should be one’s HDL cholesterol? The answer to this question is somewhat dependent on each individual’s health profile, but most data suggest that a level greater than 60 mg/dl (1.55 mmol/l) is close to optimal for most people.
The figure below (from Eckardstein, 2008; full reference at the end of this post) plots incidence of coronary events in men (on the vertical axis), over a period of 10 years, against HDL cholesterol levels (on the horizontal axis). Note: IFG = impaired fasting glucose. This relationship is similar for women, particularly post-menopausal women. Pre-menopausal women usually have higher HDL cholesterol levels than men, and a low incidence of coronary events.
From the figure above, one can say that a diabetic man with about 55 mg/dl of HDL cholesterol will have approximately the same chance, on average, of having a coronary event (a heart attack) as a man with no risk factors and about 20 mg/dl of HDL cholesterol. That chance will be about 7 percent. With 20 mg/dl of HDL cholesterol, the chance of a diabetic man having a coronary event would approach 50 percent.
We can also conclude from the figure above that a man with no risk factors will have a 5 percent chance of having a coronary event if his HDL cholesterol is about 25 mg/dl; and about 2 percent if his HDL cholesterol is greater than 60 mg/dl. This a 60 percent reduction in risk, a risk that was low to start with because of the absence of risk factors.
HDL cholesterol levels greater than 60 are associated with significantly reduced risks of coronary events, particularly for those with diabetes (the graph does not take diabetes type into consideration). Much higher levels of HDL cholesterol (beyond 60) do not seem to be associated with much lower risk of coronary events.
Conversely, a very low HDL cholesterol level (below 25) is a major risk factor when other risk factors are also present, particularly: diabetes, hypertension (high blood pressure), and familial hypercholesteromia (gene-induced very elevated LDL cholesterol).
It is not yet clear whether HDL cholesterol is a cause of reduced cardiovascular disease, or just a marker of other health factors that lead to reduced risk for cardiovascular disease. Much of the empirical evidence suggests a causal relationship, and if this is the case then it may be a good idea to try to increase HDL levels. Even if HDL cholesterol is just a marker, the same strategy that increases it may also have a positive impact on the real causative factor of which HDL cholesterol is a marker.
What can one do to increase his or her HDL cholesterol? One way is to replace refined carbs and sugars with saturated fat and cholesterol in one’s diet. (I know that this sounds counterintuitive, but seems to work.) Another is to increase one’s vitamin D status, through sun exposure or supplementation.
Other therapeutic interventions can also be used to increase HDL; some more natural than others. The figure below (also from Eckardstein, 2008) shows the maximum effects of several therapeutic interventions to increase HDL cholesterol.
Among the therapeutic interventions shown in the figure above, taking nicotinic acid (niacin) in pharmacological doses, of 1 to 3 g per day (higher dosages may be toxic), is by far the most effective way of increasing one’s HDL cholesterol. Only the niacin that causes flush is effective in this respect. No-flush niacin preparations may have some anti-inflammatory effects, but do not cause increases in HDL cholesterol.
Rimonabant, which is second to niacin in its effect on HDL cholesterol, is an appetite suppressor that has been associated with serious side effects and, to be best of my knowledge, has been largely banned from use in pharmaceutical drugs.
Third in terms of effectiveness, among the factors shown in the figure, is moderate alcohol consumption. Running about 19 miles per week (2.7 miles per day) and taking fibrates are tied in forth place.
Many people think that they are having a major allergic reaction, and have a panic attack, when they experience the niacin flush. This usually happens several minutes after taking niacin, and depends on the dose and whether niacin was consumed with food or not. It is not uncommon for one’s entire torso to turn hot red, as though the person had had major sunburn. This reaction is harmless, and usually disappears after several minutes.
One could say that, with niacin: no “pain” (i.e., flush), no gain.
Reference:
von Eckardstein, A. (2008). HDL – a difficult friend. Drug Discovery Today: Disease Mechanisms, 5(3), 315-324.
Tuesday, February 16, 2010
Large LDL and small HDL particles: The best combination
High-density lipoprotein (HDL) is one of the five main types of lipoproteins found in circulation, together with very low-density lipoprotein (VLDL), intermediate-density lipoprotein (IDL), low-density lipoprotein (LDL), and chylomicrons.
After a fatty meal, the blood is filled with chylomicrons, which carry triglycerides (TGAs). The TGAs are transferred to cells from chylomicrons via the activity of enzymes, in the form of free fatty acids (FFAs), which are used by those cells as sources of energy.
After delivering FFAs to the cells, the chylomicrons progressively lose their TGA content and “shrink”, eventually being absorbed and recycled by the liver. The liver exports part of the TGAs that it gets from chylomicrons back to cells for use as energy as well, now in the form of VLDL. As VLDL particles deliver TGAs to the cells they shrink in size, similarly to chylomicrons. As they shrink, VLDL particles first become IDL and then LDL particles.
The figure below (click on it to enlarge), from Elliott & Elliott (2009; reference at the end of this post), shows, on the same scale: (a) VLDL particles, (b) chylomicrons, (c) LDL particles, and (d) HDL particles. The dark bar at the bottom of each shot is 1000 A in length, or 100 nm (A = angstrom; nm = nanometer; 1 nm = 10 A).
As you can see from the figure, most of the LDL particles shown are about 1/4 of the length of the dark bar in diameter, often slightly more, or about 25-27 nm in size. They come in different sizes, with sizes in this range being the most common. The smaller and denser they are, the more likely they are to contribute to the formation of atherosclerotic plaque in the presence of other factors, such as chronic inflammation. The larger they become, which usually happens in diets high in saturated fat, the less likely they are to form plaque.
Note that the HDL particles are rather small compared to the LDL particles. Shouldn’t they cause plaque then? Not really. Apparently they have to be small, compared to LDL particles, to do their job effectively.
HDL is a completely different animal from VLDL, IDL and LDL. HDL particles are produced by the liver as dense disk-like particles, known as nascent HDL particles. These nascent HDL particles progressively pick up cholesterol from cells, as well as performing a number of other functions, and “fatten up” with cholesterol in the process.
This process also involves HDL particles picking up cholesterol from plaque in the artery walls, which is one of the reasons why HDL cholesterol is informally called “good” cholesterol. In fact, neither HDL nor LDL are really cholesterol; HDL and LDL are particles that carry cholesterol, protein and fat.
As far as particle size is concerned, LDL and HDL are opposites. Large LDL particles are the least likely to cause plaque formation, because LDL particles have to be approximately 25 nm in diameter or smaller to penetrate the artery walls. With HDL the opposite seems to be true, as HDL particles need to be small (compared with LDL particles) to easily penetrate the artery walls in order to pick up cholesterol, leave the artery walls with their cargo, and have it returned back to the liver.
Interestingly, some research suggests HDL particles that are larger in size, when compared with other HDL particles (not with LDL particles), seem to do a better job than very small HDL particles in terms of reducing risk of cardiovascular disease. It is also possible that a high number of larger HDL particles in the blood is indicative of elevated levels of "effective" HDL particles; i.e., particles that are effective at picking up cholesterol from the artery walls in the first place.
Another interesting aspect of this cycle is that the return to the liver of cholesterol picked up by HDL appears to be done largely via IDL and LDL particles (Elliott & Elliott, 2009), which get the cholesterol directly from HDL particles! Life is not that simple.
Reference:
William H. Elliott & Daphne C. Elliott (2009). Biochemistry and Molecular Biology. 4th Edition. New York: NY: Oxford University Press.
After a fatty meal, the blood is filled with chylomicrons, which carry triglycerides (TGAs). The TGAs are transferred to cells from chylomicrons via the activity of enzymes, in the form of free fatty acids (FFAs), which are used by those cells as sources of energy.
After delivering FFAs to the cells, the chylomicrons progressively lose their TGA content and “shrink”, eventually being absorbed and recycled by the liver. The liver exports part of the TGAs that it gets from chylomicrons back to cells for use as energy as well, now in the form of VLDL. As VLDL particles deliver TGAs to the cells they shrink in size, similarly to chylomicrons. As they shrink, VLDL particles first become IDL and then LDL particles.
The figure below (click on it to enlarge), from Elliott & Elliott (2009; reference at the end of this post), shows, on the same scale: (a) VLDL particles, (b) chylomicrons, (c) LDL particles, and (d) HDL particles. The dark bar at the bottom of each shot is 1000 A in length, or 100 nm (A = angstrom; nm = nanometer; 1 nm = 10 A).
As you can see from the figure, most of the LDL particles shown are about 1/4 of the length of the dark bar in diameter, often slightly more, or about 25-27 nm in size. They come in different sizes, with sizes in this range being the most common. The smaller and denser they are, the more likely they are to contribute to the formation of atherosclerotic plaque in the presence of other factors, such as chronic inflammation. The larger they become, which usually happens in diets high in saturated fat, the less likely they are to form plaque.
Note that the HDL particles are rather small compared to the LDL particles. Shouldn’t they cause plaque then? Not really. Apparently they have to be small, compared to LDL particles, to do their job effectively.
HDL is a completely different animal from VLDL, IDL and LDL. HDL particles are produced by the liver as dense disk-like particles, known as nascent HDL particles. These nascent HDL particles progressively pick up cholesterol from cells, as well as performing a number of other functions, and “fatten up” with cholesterol in the process.
This process also involves HDL particles picking up cholesterol from plaque in the artery walls, which is one of the reasons why HDL cholesterol is informally called “good” cholesterol. In fact, neither HDL nor LDL are really cholesterol; HDL and LDL are particles that carry cholesterol, protein and fat.
As far as particle size is concerned, LDL and HDL are opposites. Large LDL particles are the least likely to cause plaque formation, because LDL particles have to be approximately 25 nm in diameter or smaller to penetrate the artery walls. With HDL the opposite seems to be true, as HDL particles need to be small (compared with LDL particles) to easily penetrate the artery walls in order to pick up cholesterol, leave the artery walls with their cargo, and have it returned back to the liver.
Interestingly, some research suggests HDL particles that are larger in size, when compared with other HDL particles (not with LDL particles), seem to do a better job than very small HDL particles in terms of reducing risk of cardiovascular disease. It is also possible that a high number of larger HDL particles in the blood is indicative of elevated levels of "effective" HDL particles; i.e., particles that are effective at picking up cholesterol from the artery walls in the first place.
Another interesting aspect of this cycle is that the return to the liver of cholesterol picked up by HDL appears to be done largely via IDL and LDL particles (Elliott & Elliott, 2009), which get the cholesterol directly from HDL particles! Life is not that simple.
Reference:
William H. Elliott & Daphne C. Elliott (2009). Biochemistry and Molecular Biology. 4th Edition. New York: NY: Oxford University Press.
Saturday, February 13, 2010
Want to improve your cholesterol profile? Replace refined carbs and sugars with saturated fat and cholesterol in your diet
An interesting study by Clifton and colleagues (1998; full reference and link at the end of this post) looked at whether LDL cholesterol particle size distribution at baseline (i.e., beginning of the study) for various people was a determinant of lipid profile changes in each of two diets – one low and the other high in fat. This study highlights a few interesting points made in a previous post, which are largely unrelated to the main goal or findings of the study, but that are supported by side findings:
- As one increases dietary cholesterol and fat consumption, particularly saturated fat, circulating HDL cholesterol increases significantly. This happens whether one is taking niacin or not, although niacin seems to help, possibly as an independent (not moderating) factor. Increasing serum vitamin D levels, which can be done through sunlight exposure and supplementation, are also known to increase circulating HDL cholesterol.
- As one increases dietary cholesterol and fat consumption, particularly saturated fat, triglycerides in the fasting state (i.e., measured after a 8-hour fast) decrease significantly, particularly on a low carbohydrate diet. Triglycerides in the fasting state are negatively correlated with HDL cholesterol; they go down as HDL cholesterol goes up. This happens whether one is taking niacin or supplementing omega 3 fats or not, although these seem to help, possibly as independent factors.
- If one increases dietary fat intake, without also decreasing carbohydrate intake (particularly in the form of refined grains and sugars), LDL cholesterol will increase. Even so, LDL particle sizes will shift to more benign forms, which are the larger forms. Not all LDL particles change to benign forms, and there seem to be some genetic factors that influence this. LDL particles larger than 26 nm in diameter simply cannot pass through the gaps in the endothelium, which is a thin layer of cells lining the interior surface of arteries, and thus do not induce plaque formation.
The study by Clifton and colleagues (1998) involved 54 men and 51 women with a wide range of lipid profiles. They first underwent a 2-week low fat period, after which they were given two liquid supplements in addition to their low fat diet, for a period of 3 weeks. One of the liquid supplements contained 31 to 40 g of fat, and 650 to 845 mg of cholesterol. The other was fat and cholesterol free.
Studies that adopt a particular diet at baseline have the advantage of departing from a uniform diet across conditions. They also typically have one common characteristic: the baseline diet reflects the beliefs of the authors about what an ideal diet is. That is not always the case, of course. If this was indeed the case here, we have a particularly interesting study, because in that case the side findings discussed below contradicted the authors’ beliefs.
The table below shows the following measures for the participants in the study: age, body mass index (BMI), waist-to-hip ratio (WHR), total cholesterol, triglycerides, low-density lipoprotein (LDL) cholesterol, and three subtypes of high-density lipoprotein (HDL) cholesterol. LDL cholesterol is the colloquially known as the “bad” type, and “HDL” as the good one (which is an oversimplification). In short, the participants were overweight, middle-aged men and women, with relatively poor lipid profiles.
At the bottom of the table is the note “P < 0.001”, following a small “a”. This essentially means that on the rows indicated by an “a”, like the “WHR” row, the difference in the averages (e.g., 0.81 for women, and 0.93 for men, in the WHR row) was significantly different from what one would expect it to be due to chance alone. More precisely, the likelihood that the difference was due to chance was lower than 0.001, or 0.1 percent, in the case of a P < 0.001. Usually a difference between averages (a.k.a. means) associated with a P < 0.05 will be considered statistically significant.
Since the LDL cholesterol concentrations (as well as other lipoprotein concentrations) are listed on the table in mmol/L, and many people receive those measures in mg/dL in blood lipid profile test reports, below is a conversion table for LDL cholesterol (from: Wikipedia).
The table below shows the dietary intake in the low and high fat diets. Note that in the high fat diet, not only is the fat intake higher, but so is the cholesterol intake. The latter is significantly higher, more than 4 times the intake in the low fat diet, and about 2.5 times the recommended daily value by the U.S. Food and Drug Administration. The total calorie intake is reported as slightly lower in the high fat diet than in the low fat diet.
Note that the largest increase was in saturated fat, followed by an almost equally large increase in monounsaturated fat. This, together with the increase in cholesterol, mimics a move to a diet where fatty meat and organs are consumed in higher quantities, with a corresponding reduction in the intake of refined carbohydrates (e.g., bread, pasta, sugar, potatoes) and lean meats.
Finally, the table below shows the changes in lipid profiles in the low and high fat diets. Note that all subtypes of HDL (or "good") cholesterol concentrations were significantly higher in the high fat diet, which is very telling, because HDL cholesterol concentrations are much better predictors of cardiovascular disease than LDL or total cholesterol concentrations. The higher the HDL cholesterol, the lower the risk of cardiovascular disease.
In the table above, we also see that triglycerides are significantly lower in the high fat diet, which is also good, because high fasting triglyceride concentrations are associated with cardiovascular disease and also insulin resistance (which is associated with diabetes).
However, the total and LDL cholesterol were also significantly higher in the high fat compared to the low fat diet. Is this as bad as it sounds? Not when we look at other factors that are not clear from the tables in the article.
One of those factors is the likely change in LDL particle size. LDL particle sizes almost always increase with significant increases in HDL; frequently going up in diameter beyond 26 nm, and thus passing the threshold beyond which an LDL particle can penetrate the endothelium and help form a plaque.
Another important factor to take into consideration is the somewhat strange decision by the authors to use the Friedewald equation to estimate the LDL concentrations in the low and high fat diets. Through the Friedewald equation, LDL is calculated as follows (where TC is total cholesterol):
LDL = TC – HDL – Triglycerides / 5
Here is one of the problems with the Friedewald equation. Let us assume that an individual has the following lipid profile numbers: TC = 200, HDL = 50, and trigs. = 150. The calculated LDL will be 120. Let us assume that this same individual reduces trigs. to 50, from the previous 150, keeping all of the other measures constant. This is a major improvement. Yet, the calculated LDL will now be 140, and a doctor will tell this person to consider taking statins!
By the way, most people who do a blood test and get their lipid profile report also get their LDL calculated through the Friedewald equation. Usually this is indicated through a "CALC" note next to the description of the test or the calculated LDL number.
Finally, total cholesterol is not a very useful measure, because an elevated total cholesterol may be primarily reflecting an elevated HDL, which is healthy. Also, a slightly elevated total cholesterol seems to be protective, as it is associated with reduced overall mortality and also reduced mortality from cardiovascular disease, according to U-curve regression studies comparing mortality and total cholesterol levels in different countries.
We do not know for sure that the participants in this study were consuming a lot of refined carbohydrates and/or sugars at baseline. But it is a safe bet that they were, since they were consuming 214 g of carbohydrates per day. It is difficult, although not impossible, to eat that many carbohydrates per day by eating only vegetables and fruits, which are mostly water.
This is why when one goes on a paleo diet, he or she reduces significantly the amount of dietary carbohydrates; even more so on a targeted low carbohydrate diet, such as the Atkins diet. Richard K. Bernstein, who is a type 1 diabetic and has been adopting a strict low carbohydrate diet during most of his adult life, had the following lipid profile at 72 years of age: HDL = 118, LDL = 53, trigs. = 45. His fasting blood sugar was reportedly 83 mg/dl. Click here to listen to an interview with Dr. Bernstein on the The Livin' La Vida Low-Carb Show.
The lipid profile improvement observed (e.g., a 14 percent increase in HDL from baseline for men, and about half that for women, in only 3 weeks) was very likely due to an increase in dietary saturated fat and cholesterol combined with a decrease in refined carbohydrates and sugars. The improvement would probably have been even more impressive with a higher increase in saturated fat, as long as it was accompanied by the elimination of refined carbohydrates and sugars from the participants’ diets.
Reference:
Clifton, P. M., M. Noakes, and P. J. Nestel (1998). LDL particle size and LDL and HDL cholesterol changes with dietary fat and cholesterol in healthy subjects. J. Lipid. Res. 39: 1799–1804.
- As one increases dietary cholesterol and fat consumption, particularly saturated fat, circulating HDL cholesterol increases significantly. This happens whether one is taking niacin or not, although niacin seems to help, possibly as an independent (not moderating) factor. Increasing serum vitamin D levels, which can be done through sunlight exposure and supplementation, are also known to increase circulating HDL cholesterol.
- As one increases dietary cholesterol and fat consumption, particularly saturated fat, triglycerides in the fasting state (i.e., measured after a 8-hour fast) decrease significantly, particularly on a low carbohydrate diet. Triglycerides in the fasting state are negatively correlated with HDL cholesterol; they go down as HDL cholesterol goes up. This happens whether one is taking niacin or supplementing omega 3 fats or not, although these seem to help, possibly as independent factors.
- If one increases dietary fat intake, without also decreasing carbohydrate intake (particularly in the form of refined grains and sugars), LDL cholesterol will increase. Even so, LDL particle sizes will shift to more benign forms, which are the larger forms. Not all LDL particles change to benign forms, and there seem to be some genetic factors that influence this. LDL particles larger than 26 nm in diameter simply cannot pass through the gaps in the endothelium, which is a thin layer of cells lining the interior surface of arteries, and thus do not induce plaque formation.
The study by Clifton and colleagues (1998) involved 54 men and 51 women with a wide range of lipid profiles. They first underwent a 2-week low fat period, after which they were given two liquid supplements in addition to their low fat diet, for a period of 3 weeks. One of the liquid supplements contained 31 to 40 g of fat, and 650 to 845 mg of cholesterol. The other was fat and cholesterol free.
Studies that adopt a particular diet at baseline have the advantage of departing from a uniform diet across conditions. They also typically have one common characteristic: the baseline diet reflects the beliefs of the authors about what an ideal diet is. That is not always the case, of course. If this was indeed the case here, we have a particularly interesting study, because in that case the side findings discussed below contradicted the authors’ beliefs.
The table below shows the following measures for the participants in the study: age, body mass index (BMI), waist-to-hip ratio (WHR), total cholesterol, triglycerides, low-density lipoprotein (LDL) cholesterol, and three subtypes of high-density lipoprotein (HDL) cholesterol. LDL cholesterol is the colloquially known as the “bad” type, and “HDL” as the good one (which is an oversimplification). In short, the participants were overweight, middle-aged men and women, with relatively poor lipid profiles.
At the bottom of the table is the note “P < 0.001”, following a small “a”. This essentially means that on the rows indicated by an “a”, like the “WHR” row, the difference in the averages (e.g., 0.81 for women, and 0.93 for men, in the WHR row) was significantly different from what one would expect it to be due to chance alone. More precisely, the likelihood that the difference was due to chance was lower than 0.001, or 0.1 percent, in the case of a P < 0.001. Usually a difference between averages (a.k.a. means) associated with a P < 0.05 will be considered statistically significant.
Since the LDL cholesterol concentrations (as well as other lipoprotein concentrations) are listed on the table in mmol/L, and many people receive those measures in mg/dL in blood lipid profile test reports, below is a conversion table for LDL cholesterol (from: Wikipedia).
The table below shows the dietary intake in the low and high fat diets. Note that in the high fat diet, not only is the fat intake higher, but so is the cholesterol intake. The latter is significantly higher, more than 4 times the intake in the low fat diet, and about 2.5 times the recommended daily value by the U.S. Food and Drug Administration. The total calorie intake is reported as slightly lower in the high fat diet than in the low fat diet.
Note that the largest increase was in saturated fat, followed by an almost equally large increase in monounsaturated fat. This, together with the increase in cholesterol, mimics a move to a diet where fatty meat and organs are consumed in higher quantities, with a corresponding reduction in the intake of refined carbohydrates (e.g., bread, pasta, sugar, potatoes) and lean meats.
Finally, the table below shows the changes in lipid profiles in the low and high fat diets. Note that all subtypes of HDL (or "good") cholesterol concentrations were significantly higher in the high fat diet, which is very telling, because HDL cholesterol concentrations are much better predictors of cardiovascular disease than LDL or total cholesterol concentrations. The higher the HDL cholesterol, the lower the risk of cardiovascular disease.
In the table above, we also see that triglycerides are significantly lower in the high fat diet, which is also good, because high fasting triglyceride concentrations are associated with cardiovascular disease and also insulin resistance (which is associated with diabetes).
However, the total and LDL cholesterol were also significantly higher in the high fat compared to the low fat diet. Is this as bad as it sounds? Not when we look at other factors that are not clear from the tables in the article.
One of those factors is the likely change in LDL particle size. LDL particle sizes almost always increase with significant increases in HDL; frequently going up in diameter beyond 26 nm, and thus passing the threshold beyond which an LDL particle can penetrate the endothelium and help form a plaque.
Another important factor to take into consideration is the somewhat strange decision by the authors to use the Friedewald equation to estimate the LDL concentrations in the low and high fat diets. Through the Friedewald equation, LDL is calculated as follows (where TC is total cholesterol):
LDL = TC – HDL – Triglycerides / 5
Here is one of the problems with the Friedewald equation. Let us assume that an individual has the following lipid profile numbers: TC = 200, HDL = 50, and trigs. = 150. The calculated LDL will be 120. Let us assume that this same individual reduces trigs. to 50, from the previous 150, keeping all of the other measures constant. This is a major improvement. Yet, the calculated LDL will now be 140, and a doctor will tell this person to consider taking statins!
By the way, most people who do a blood test and get their lipid profile report also get their LDL calculated through the Friedewald equation. Usually this is indicated through a "CALC" note next to the description of the test or the calculated LDL number.
Finally, total cholesterol is not a very useful measure, because an elevated total cholesterol may be primarily reflecting an elevated HDL, which is healthy. Also, a slightly elevated total cholesterol seems to be protective, as it is associated with reduced overall mortality and also reduced mortality from cardiovascular disease, according to U-curve regression studies comparing mortality and total cholesterol levels in different countries.
We do not know for sure that the participants in this study were consuming a lot of refined carbohydrates and/or sugars at baseline. But it is a safe bet that they were, since they were consuming 214 g of carbohydrates per day. It is difficult, although not impossible, to eat that many carbohydrates per day by eating only vegetables and fruits, which are mostly water.
This is why when one goes on a paleo diet, he or she reduces significantly the amount of dietary carbohydrates; even more so on a targeted low carbohydrate diet, such as the Atkins diet. Richard K. Bernstein, who is a type 1 diabetic and has been adopting a strict low carbohydrate diet during most of his adult life, had the following lipid profile at 72 years of age: HDL = 118, LDL = 53, trigs. = 45. His fasting blood sugar was reportedly 83 mg/dl. Click here to listen to an interview with Dr. Bernstein on the The Livin' La Vida Low-Carb Show.
The lipid profile improvement observed (e.g., a 14 percent increase in HDL from baseline for men, and about half that for women, in only 3 weeks) was very likely due to an increase in dietary saturated fat and cholesterol combined with a decrease in refined carbohydrates and sugars. The improvement would probably have been even more impressive with a higher increase in saturated fat, as long as it was accompanied by the elimination of refined carbohydrates and sugars from the participants’ diets.
Reference:
Clifton, P. M., M. Noakes, and P. J. Nestel (1998). LDL particle size and LDL and HDL cholesterol changes with dietary fat and cholesterol in healthy subjects. J. Lipid. Res. 39: 1799–1804.
Subscribe to:
Posts (Atom)